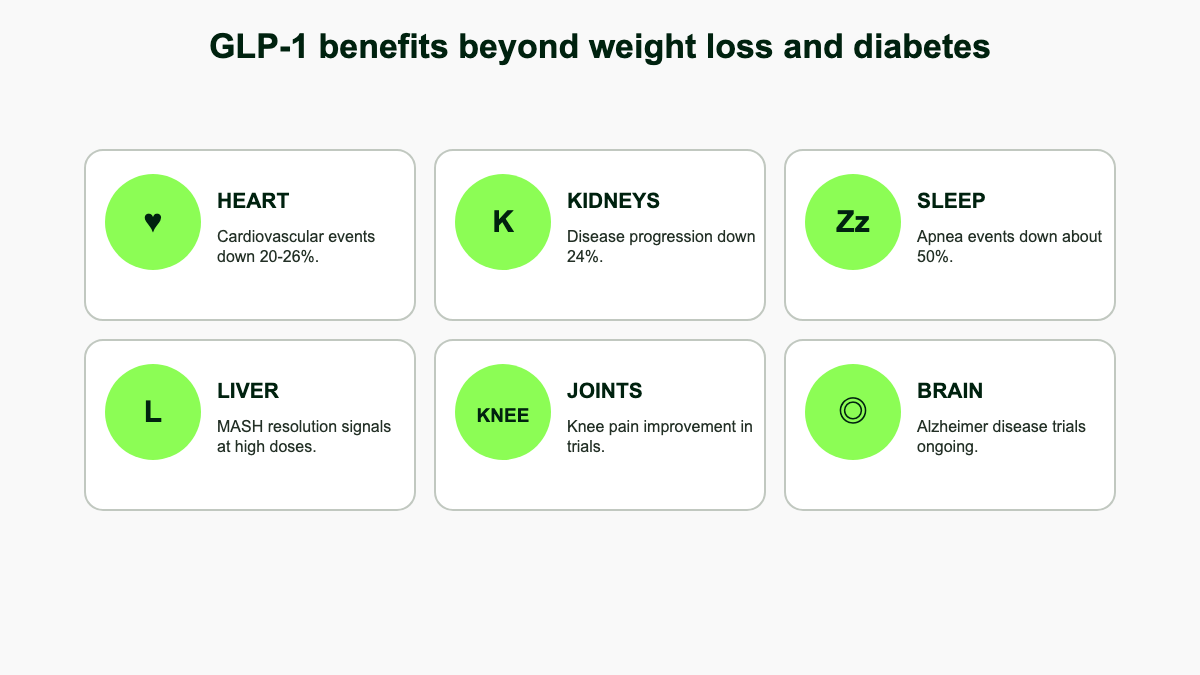
GLP-1 Benefits & Results
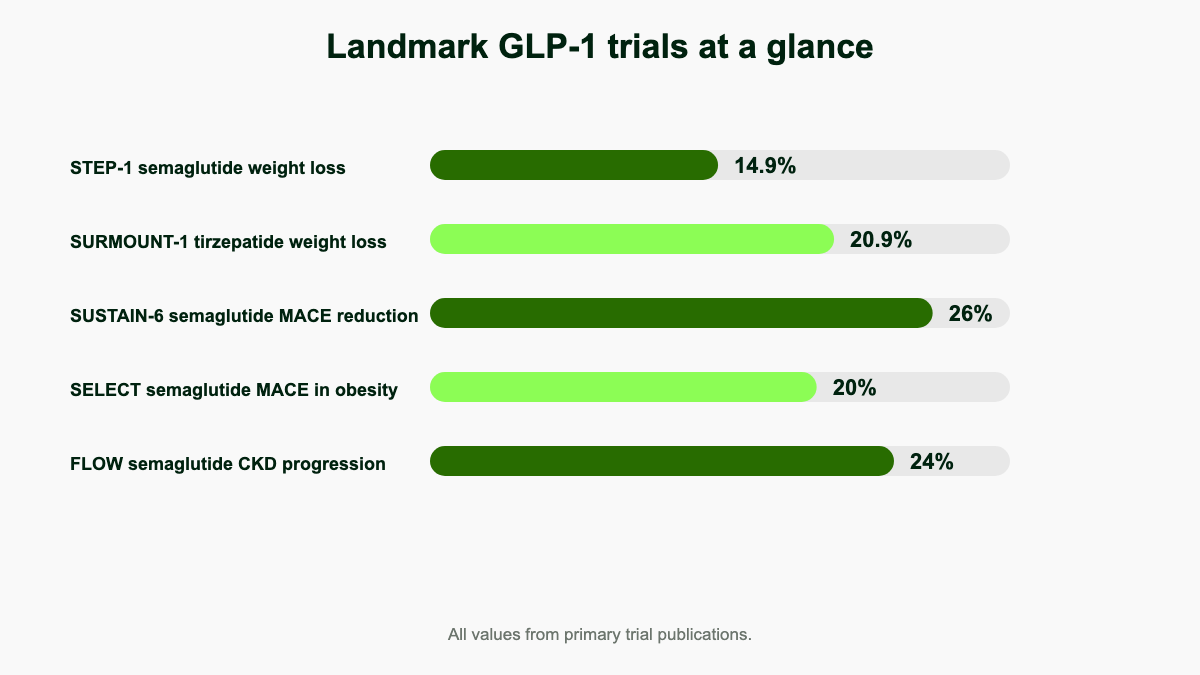
Summary: In randomized trials GLP-1 medications produced about 14.9% body weight loss with semaglutide 2.4 mg over 68 weeks (STEP-1) and up to 20.9% with tirzepatide 15 mg over 72 weeks (SURMOUNT-1), alongside a 20% reduction in major cardiovascular events (SELECT), a 24% reduction in kidney disease progression in type 2 diabetes (FLOW), roughly 60% lower apnea-hypopnea index in obstructive sleep apnea (SURMOUNT-OSA), and biopsy-confirmed MASH resolution in 63% of patients (ESSENCE). [1][2][3][4][5][6]
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.
Weight loss results: how much, how fast, and which drug
The STEP-1 trial randomized 1,961 adults with overweight or obesity (without diabetes) to once-weekly semaglutide 2.4 mg or placebo. Mean change in body weight from baseline to week 68 was −14.9% with semaglutide versus −2.4% with placebo, an estimated treatment difference of about 12.4 percentage points. About 86% of semaglutide-treated participants lost at least 5% of body weight, 69% lost at least 10%, and 50% lost at least 15%. Roughly one in three reached the 20% threshold that historically required bariatric surgery. [1]
SURMOUNT-1 evaluated tirzepatide, a dual GIP/GLP-1 receptor agonist, in 2,539 adults with obesity. At 72 weeks the mean weight reduction was 15.0% on 5 mg, 19.5% on 10 mg, and 20.9% on 15 mg, compared with 3.1% on placebo. At the top dose, 57% of participants reached at least 20% weight loss and 36% reached at least 25%, putting average response into the range typically seen with sleeve gastrectomy. [2] In the head-to-head SURMOUNT-5 phase 3b trial published in NEJM in May 2025, tirzepatide produced a mean 20.2% weight reduction versus 13.7% with semaglutide 2.4 mg over 72 weeks in adults with obesity but without diabetes, along with a waist-circumference reduction of 18.4 cm versus 13.0 cm and lower GI-related discontinuation (2.7% versus 5.6%). [7]
Earlier-generation liraglutide 3.0 mg (SCALE program) typically delivers 5%–8% mean weight loss over 56 weeks, well below the newer agents. Oral semaglutide 25 mg dosed for obesity produced about 13.6% weight loss over 64–68 weeks in OASIS-4, narrowing the injectable-versus-oral gap. [8] At the experimental edge, the triple GIP/GLP-1/glucagon receptor agonist retatrutide produced a least-squares mean −24.2% weight reduction at 12 mg over 48 weeks in a phase 2 trial of 338 adults, compared with −2.1% for placebo, with 100% of completers in the highest-dose group losing at least 5% and 83% losing at least 15%. [9] Phase 3 retatrutide trials in the TRIUMPH program are underway and could push average weight loss further toward gastric-bypass territory.
Speed matters less than most patients expect. In STEP-1, weight curves separated within four weeks but did not plateau until roughly week 60, and the SURMOUNT-1 plateau arrived around week 64. Patients who lose 5% by week 16 are good candidates to continue; non-responders at that mark rarely catch up without dose escalation or switch.
Long-term durability: what happens past one year
STEP 5 extended treatment with semaglutide 2.4 mg to 104 weeks in 304 adults. Mean weight change at week 104 was −15.2% with semaglutide versus −2.6% with placebo, an estimated treatment difference of −12.6 percentage points. Critically, weight loss was maintained between weeks 52 and 104 with no meaningful additional drop or regain on therapy. About 77% of semaglutide-treated participants remained at or below the 5% weight-loss threshold at two years versus 34% on placebo. [10] Real-world durability tracks the trial closely so long as patients stay on dose; long-term observational cohorts of liraglutide and dulaglutide show similar maintenance.
The picture changes the day the prescription stops. In the STEP-1 extension, participants who discontinued semaglutide regained roughly two-thirds of lost weight by week 120, with concurrent rebound in waist circumference, blood pressure, lipids, A1c, and CRP. [11] This pattern parallels what happens after stopping antihypertensives or statins; obesity behaves like a chronic disease, and removing the medication that controls it returns physiology toward baseline. Discussions about “tapering off” should account for this biology rather than assume motivation alone will hold weight down.
How GLP-1 medications drive these results
Native glucagon-like peptide-1 is a 30-amino-acid hormone secreted by L-cells in the distal small intestine within minutes of a meal. It potentiates glucose-dependent insulin secretion, suppresses glucagon, slows gastric emptying, and signals satiety in the hypothalamus and brainstem. Native GLP-1 has a half-life of about two minutes because of rapid degradation by dipeptidyl peptidase-4. Modern GLP-1 receptor agonists are engineered to resist DPP-4 cleavage and bind albumin so they persist for days; semaglutide has a half-life of about a week, supporting once-weekly dosing. Tirzepatide adds glucose-dependent insulinotropic polypeptide (GIP) receptor agonism on the same molecular backbone, layering complementary insulinotropic and adipocyte effects on top of the GLP-1 mechanism. Retatrutide adds a third axis, glucagon receptor agonism, which appears to drive additional energy expenditure and hepatic fat clearance. The pharmacology explains why each successive class member tends to deliver larger average weight loss than the last.
Beyond the gut and pancreas, GLP-1 receptors are expressed in the heart, kidneys, vasculature, brainstem, hypothalamus, hippocampus, and mesolimbic reward circuits. This anatomy frames why one drug class touches so many organs at once. Mechanistic biopsies and imaging substudies report decreased macrophage infiltration in atherosclerotic plaque, lower oxidative stress in the kidney, reduced hepatic de novo lipogenesis, and lowered reward-circuit reactivity to food cues, each of which maps to a documented clinical benefit.
Glycemic control in type 2 diabetes
In the SUSTAIN program, semaglutide 1.0 mg lowered HbA1c by roughly 1.5–1.8 percentage points versus comparators including sitagliptin, exenatide ER, and insulin glargine, with concurrent weight loss of 4.5–6.5 kg. [12] Tirzepatide pushed those numbers further. SURPASS-2 directly compared three doses of tirzepatide against semaglutide 1 mg in 1,879 adults on metformin. After 40 weeks the semaglutide arm reached an A1c of 6.42%, while tirzepatide 5 mg, 10 mg, and 15 mg reached 6.19%, 5.91%, and 5.82% respectively. Tirzepatide 15 mg also produced 12.4 kg of weight loss compared with 6.2 kg on semaglutide 1 mg. [13] Across SURPASS-1 through SURPASS-5, the tirzepatide 15 mg dose produced HbA1c reductions of approximately 2.0–2.6 percentage points, with a substantial share of patients reaching A1c below 5.7%, the non-diabetes range, on monotherapy.
Hypoglycemia rates remain low when GLP-1 therapy is used without insulin or sulfonylureas, because GLP-1 signaling stimulates insulin only in the presence of elevated glucose. The American Diabetes Association Standards of Care list GLP-1 receptor agonists or GLP-1/GIP co-agonists as preferred injectable therapies in type 2 diabetes when weight management or cardiovascular risk reduction are priorities, and the 2024 update places them ahead of basal insulin in most patients without contraindications. [14] Beta-cell function metrics (HOMA-B, proinsulin/insulin ratio) also improve in long-term follow-up, hinting at a disease-modifying signal that older drug classes did not deliver.
Cardiovascular outcomes across diabetes and obesity
SUSTAIN-6 enrolled 3,297 adults with type 2 diabetes at high cardiovascular risk and randomized them to subcutaneous semaglutide or placebo for 104 weeks. The primary composite of cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke occurred in 6.6% of semaglutide patients versus 8.9% on placebo, a hazard ratio of 0.74 (95% CI 0.58–0.95), corresponding to a 26% relative risk reduction. Nonfatal stroke was reduced by 39%. [15]
LEADER followed 9,340 patients with type 2 diabetes for a median 3.8 years on liraglutide 1.8 mg or placebo. The primary three-point MACE endpoint occurred in 13.0% of liraglutide patients versus 14.9% on placebo (HR 0.87; 95% CI 0.78–0.97), with cardiovascular death lower by 22%. [16] REWIND extended the cardiovascular signal to a primary-prevention population: 9,901 adults with type 2 diabetes (69% without prior CV disease) on dulaglutide 1.5 mg saw a 12% relative reduction in MACE over a median 5.4 years (HR 0.88; 95% CI 0.79–0.99). [17]
The landmark SELECT trial extended these findings outside diabetes. Among 17,604 adults aged 45 or older with overweight or obesity and established cardiovascular disease but without diabetes, semaglutide 2.4 mg cut the primary MACE composite by 20% over a mean 39.8 months (HR 0.80; 95% CI 0.72–0.90; p<0.001). [4] Effects emerged before significant weight loss had accumulated, suggesting weight-independent vascular pathways. On the strength of SELECT, the FDA expanded Wegovy's label in March 2024 to include reducing the risk of cardiovascular death, heart attack, and stroke in adults with cardiovascular disease and overweight or obesity.
Oral semaglutide passed the same bar in two trials. PIONEER 6 demonstrated cardiovascular safety (HR 0.79 for MACE, noninferior to placebo) in 3,183 high-risk patients with type 2 diabetes. [18] SOUL, published in NEJM in March 2025, then established superiority: among 9,650 adults with type 2 diabetes plus established atherosclerotic cardiovascular disease, chronic kidney disease, or both, oral semaglutide 14 mg cut MACE by 14% versus placebo (12.0% vs 13.8%; HR 0.86; 95% CI 0.77–0.96; p=0.006), with no excess in serious adverse events. [19] The SOUL data closes the loop showing that the cardiovascular signal does not depend on the injectable formulation.
Kidney protection: the FLOW trial
FLOW enrolled 3,533 patients with type 2 diabetes and chronic kidney disease (eGFR 50–75 with UACR >300, or eGFR 25–<50 with UACR >100). The trial was stopped early for efficacy. The primary composite (kidney failure, sustained ≥50% eGFR decline, or kidney/cardiovascular death) occurred in 18.7% of semaglutide patients versus 23.2% on placebo, a hazard ratio of 0.76, equating to a 24% relative risk reduction. The mean annual decline in eGFR was 1.16 mL/min/1.73 m² slower with semaglutide (95% CI 0.86–1.47; p<0.001). All-cause mortality dropped 20% (HR 0.80; 95% CI 0.67–0.95) and cardiovascular death fell 29% (HR 0.71; 95% CI 0.56–0.89). [5] In January 2025 the FDA approved Ozempic to reduce the risk of worsening kidney disease and cardiovascular death in adults with type 2 diabetes and CKD. The benefit held whether or not patients were on an SGLT2 inhibitor, suggesting GLP-1 and SGLT2 mechanisms are additive rather than redundant.
Heart failure with preserved ejection fraction
HFpEF is the dominant heart failure phenotype in obese patients and historically has had few effective therapies. STEP-HFpEF randomized 529 patients with EF ≥45% and BMI ≥30 to semaglutide 2.4 mg or placebo for 52 weeks. The Kansas City Cardiomyopathy Questionnaire Clinical Summary Score (KCCQ-CSS) improved by 16.6 points with semaglutide versus 8.7 points with placebo. Six-minute walk distance improved by 17.1 m more with semaglutide, CRP fell, NT-proBNP dropped, and serious adverse events were roughly halved (161 vs 301). [20]
SUMMIT extended this to tirzepatide. In 731 patients with HFpEF and obesity, tirzepatide 15 mg cut the composite of cardiovascular death or worsening heart-failure events by roughly 38% over a median 2 years; worsening HF events occurred in 8.0% of the tirzepatide arm versus 14.2% on placebo. KCCQ-CSS improved by 19.5 points versus 12.7. CMR substudies showed reductions in left-ventricular mass and paracardiac adipose tissue, indicating tirzepatide remodels the heart, not just the scale. [21] Together STEP-HFpEF and SUMMIT mark the first medications with a meaningful HFpEF outcome signal in patients with obesity.
Sleep apnea: SURMOUNT-OSA
SURMOUNT-OSA tested tirzepatide in 469 adults with moderate-to-severe obstructive sleep apnea and obesity across two parallel trials, one without and one with PAP therapy. Tirzepatide reduced the apnea-hypopnea index by about 27.4 events/hour in the no-PAP trial and 30.4 events/hour in the on-PAP trial, corresponding to roughly 55%–63% relative reductions versus placebo. About half of treated patients dropped to mild or no OSA. Body weight, hypoxic burden, hsCRP, and systolic blood pressure also improved. [6] In December 2024 the FDA approved Zepbound for moderate-to-severe OSA in adults with obesity, the first drug indication for OSA. For patients who struggle to tolerate CPAP, this opened a route that earlier weight-loss medications never offered.
Liver disease: MASH and MASLD
In MASH (metabolic dysfunction-associated steatohepatitis, formerly NASH), the phase 3 ESSENCE trial of semaglutide 2.4 mg in 800 patients showed resolution of steatohepatitis without worsening of fibrosis in 62.9% of patients versus 34.3% on placebo at 72 weeks, with improvement in liver fibrosis in 36.8% versus 22.4%. ALT, AST, and noninvasive fibrosis markers (FIB-4, ELF) all improved. [22] The phase 2 SYNERGY-NASH trial of tirzepatide in 190 patients with biopsy-proven F2/F3 fibrosis showed even higher MASH resolution rates: 51.8%, 62.8%, and 73.3% on tirzepatide 5, 10, and 15 mg versus 13.2% on placebo at 52 weeks, with one-stage fibrosis improvement in 53%–59% of treated patients versus 33% on placebo. [23] Phase 3 tirzepatide MASH trials are ongoing. For patients with MASLD without overt steatohepatitis, weight loss alone usually drives meaningful liver-fat reduction, and MR-PDFF substudies in SURMOUNT and STEP show 40%–60% relative declines in hepatic steatosis.
Knee osteoarthritis: STEP 9
STEP 9 randomized 407 adults with obesity and moderate-to-severe knee osteoarthritis to semaglutide 2.4 mg or placebo for 68 weeks. Mean weight loss was 13.7% versus 3.2%. The WOMAC pain score improved by 41.7 points with semaglutide versus 27.5 points on placebo, a clinically meaningful 14.1-point treatment difference. Physical-function scores and analgesic use improved in parallel. [24] Imaging substudies have hinted at small increases in cartilage thickness and reductions in joint-space inflammation, though dedicated structural trials are still needed. For patients facing knee replacement, semaglutide now sits in the conversation alongside physical therapy and intra-articular injections.
Brain, addiction, and behavior signals
GLP-1 receptors are expressed in mesolimbic reward circuits, which has driven interest in addiction indications. A 2023 prospective cohort by Quddos and colleagues followed 153 adults on semaglutide or tirzepatide and reported significant reductions in self-reported drinks per day (−1.31 for semaglutide, −1.54 for tirzepatide; both p ≤ 0.001) and binge episodes (−2.05 and −3.8) versus matched controls. [25] A 2024 randomized trial of low-dose semaglutide in alcohol use disorder showed reductions in heavy drinking days and craving scores. Pharmacovigilance studies and electronic health record analyses report consistent reductions in alcohol-related healthcare utilization among GLP-1 users. NIH and Yale-led trials of semaglutide for AUD are underway.
Smoking cessation signals are smaller but consistent. A randomized trial of dulaglutide as adjunct to varenicline showed similar abstinence rates with significantly less post-cessation weight gain. Anecdotal reports of reduced nicotine, gambling, and compulsive shopping behaviors have driven interest, though randomized data remain thin and the existing trials are short and small.
For Alzheimer disease, the picture is mixed. The phase 3 evoke and evoke+ trials of oral semaglutide 14 mg in 3,800 adults with mild cognitive impairment or mild dementia due to Alzheimer disease did not show superiority over placebo on CDR-SB (Clinical Dementia Rating-Sum of Boxes) at 104 weeks. CSF biomarkers (p-tau181, p-tau217, neuroinflammation markers) did improve by up to 10%, but the biomarker shift did not translate to clinical slowing of decline. [26] Whether semaglutide and other GLP-1s prevent rather than treat dementia remains an open question, with primary-prevention cohorts still maturing.
PCOS, fertility, and women's health
Polycystic ovary syndrome is driven by insulin resistance and hyperandrogenism, both of which respond to GLP-1 therapy. Meta-analyses of small randomized trials report higher menstrual regularity, lower free testosterone, higher SHBG, and improved natural pregnancy rates with GLP-1 receptor agonists in overweight and obese women with PCOS. In one comparative study, exenatide-treated patients had a 43.6% natural pregnancy rate versus 18.7% on metformin alone. [27] GLP-1 medications are not approved for use during pregnancy or fertility treatment, and the consensus is to discontinue at least 2 months before conception attempts. Anecdotal “Ozempic baby” reports almost certainly reflect restored ovulation in women who previously had subfertility from obesity and insulin resistance.
Body composition: where the weight goes
Average weight loss numbers hide an important question: how much of the loss is fat versus lean tissue? DXA substudies of STEP and SURMOUNT report that lean soft tissue accounts for roughly 25%–40% of total weight lost on GLP-1 monotherapy, a range comparable to caloric restriction or bariatric surgery at similar magnitudes of weight loss. In STEP-1, lean mass decreased about 9.7% while fat mass dropped about 19.3%, so the proportion of body weight that is lean tissue actually rose. [28] In SURMOUNT-1, roughly 25% of weight lost was lean and 75% was fat. Real-world cohorts that pair GLP-1 therapy with structured resistance training 3–5 days per week and protein intake of 1.2–1.6 g/kg report lean-mass losses of less than 10% of total weight lost, a meaningfully better split. The implication is not to skip the medication; it is to pair it with the lifestyle inputs that protect muscle.
Tirzepatide vs semaglutide: head-to-head detail
Two trials directly compare the two leading agents. SURPASS-2 in 1,879 adults with type 2 diabetes on metformin showed tirzepatide 15 mg achieving an A1c of 5.82% versus 6.42% on semaglutide 1 mg, and 12.4 kg of weight loss versus 6.2 kg, both differences statistically significant. [13] SURMOUNT-5 in 751 adults with obesity but without diabetes showed tirzepatide producing a 20.2% mean weight reduction versus 13.7% with semaglutide 2.4 mg over 72 weeks, a 6.5 percentage-point gap, with 64.6% of tirzepatide users reaching at least 15% weight loss versus 40.1% on semaglutide. [7] On tolerability, the head-to-head data slightly favors tirzepatide: GI-related discontinuation in SURMOUNT-5 was 2.7% versus 5.6%. Real-world data echo the efficacy gap, with EHR-cohort studies reporting roughly 2-fold higher likelihood of reaching 10% weight loss on tirzepatide versus semaglutide at 12 months. The clinical implication: for many patients, tirzepatide produces more weight loss with somewhat better tolerability, though semaglutide retains the broadest cardiovascular and kidney evidence base.
Quieter food noise and quality of life
Patients on GLP-1 therapy frequently report a quieting of intrusive food thoughts, often called “food noise.” Mechanistically this combines direct GLP-1 receptor signaling in hypothalamic and brainstem appetite centers with slower gastric emptying, dopamine modulation in mesolimbic reward circuits, and reduced hedonic eating. Trial questionnaires capture the same change quantitatively. In STEP 1–4 pooled patient-reported outcomes, IWQOL-Lite-CT physical-function scores and SF-36 physical and mental component summaries all improved more with semaglutide than placebo at week 68 (p<0.001), with the largest effects in patients who lost ≥10% of body weight. [29] SURMOUNT-1 showed comparable improvements in physical functioning and self-esteem domains. Patients describe regaining the ability to climb stairs, sit comfortably, fit into airline seats, and play with grandchildren, and many rank these changes higher than the scale number itself.
Limitations and trade-offs
- Weight regain after stopping. STEP-1 extension data show roughly two-thirds of lost weight is regained by week 120 off therapy, with cardiometabolic markers drifting back toward baseline. [11] Plan for indefinite therapy or accept partial regain.
- Lean mass loss. About 25%–40% of weight lost on monotherapy comes from lean tissue. Resistance training 3 or more days per week and protein intake of 1.2–1.6 g/kg blunt this effect. [28]
- GI side effects. Nausea, vomiting, diarrhea, and constipation are the dominant reasons for discontinuation, usually concentrated in the dose-escalation phase. SURMOUNT-5 showed lower GI-related discontinuation with tirzepatide (2.7%) than semaglutide (5.6%). [7] Slower titration helps.
- Rare but important risks. Pancreatitis, gallbladder events, and worsening diabetic retinopathy have been observed; medullary thyroid carcinoma history (or MEN2) is a contraindication based on rodent data. [30]
- Cost and supply. List prices remain in the $1,000–$1,300/month range, insurance coverage for obesity (without diabetes or cardiovascular indications) is uneven, and shortages periodically interrupt supply. Compounded versions carry their own quality and regulatory questions.
- Pregnancy, planned pregnancy, breastfeeding, and severe gastroparesis. All weigh against starting or continuing therapy.
- Anesthesia and elective surgery. Delayed gastric emptying raises aspiration risk; current ASA guidance suggests holding GLP-1s before elective procedures, with timing depending on dose and indication.
Blood pressure, lipids, and inflammation
The cardiometabolic improvements on GLP-1 therapy extend beyond weight, glucose, and hard outcomes. STEP and SURMOUNT participants typically saw systolic blood pressure drop 4–7 mmHg, often allowing dose reductions of antihypertensives within the first six months. SURMOUNT-1 reported a roughly 6.2 mmHg systolic decline at 72 weeks on tirzepatide 15 mg. Lipid panels improve in parallel: triglycerides typically fall 20%–30%, LDL drops 5%–10%, and HDL rises modestly, with the largest movement in patients with metabolic syndrome at baseline. High-sensitivity CRP, a marker of low-grade inflammation, falls 25%–50% across STEP, SURMOUNT, STEP-HFpEF, and SUMMIT, and the CRP signal in SELECT decoupled from weight loss within the first weeks, supporting a direct anti-inflammatory mechanism. These changes likely contribute to the cardiovascular and kidney benefits and partially explain why outcome curves separate before substantial weight has been lost.
Stroke and peripheral artery disease
Stroke prevention is one of the most consistent vascular signals across the trial program. SUSTAIN-6 reported a 39% reduction in nonfatal stroke with semaglutide, REWIND a 24% reduction with dulaglutide, and SELECT a numerically lower stroke rate that contributed to the composite. Peripheral artery disease has its own emerging dataset. The STRIDE trial, presented at ACC.25 alongside SOUL, showed that semaglutide 1 mg increased maximum walking distance by 26 m versus placebo at 52 weeks in patients with type 2 diabetes and symptomatic PAD, with improvements in ankle-brachial index and quality-of-life scores. PAD has very few effective medications, and the STRIDE result extends the GLP-1 vascular story into the legs.
Cost, access, and the long-term decision
Wegovy, Zepbound, Ozempic, and Mounjaro carry list prices in the $1,000–$1,300 per month range in the United States, with manufacturer savings cards, employer plans, and direct-to-consumer cash programs (NovoCare, LillyDirect) reducing out-of-pocket cost to a few hundred dollars for many patients. Coverage for the obesity-only indication remains uneven and varies sharply by employer, state Medicaid program, and Medicare Part D rules; the cardiovascular and OSA labels expanded coverage materially in 2024–2025. Outside the US, list prices are typically a fraction of US pricing. Compounded versions surged during shortage periods but face contracting legal status now that brand supply has stabilized, and they bring quality-control questions that prescribers and patients should weigh carefully.
Cost-effectiveness modeling has shifted as the indication list has grown. The Mass General Brigham analysis of semaglutide for knee osteoarthritis with obesity found the drug crossed standard willingness-to-pay thresholds when downstream surgery and disability costs were modeled. ICER and similar bodies have generally found GLP-1s cost-effective in cardiovascular and CKD indications and borderline cost-effective in obesity-only use at current US list prices, with cost-effectiveness improving as prices fall through patent expiry, formulation competition, and emerging biosimilars over the next few years. Patients evaluating affordability should look at bundled outcomes (weight, A1c, BP meds dropped, insurance copays avoided) rather than the medication price line in isolation.
Mortality signals
Across the major trial program, GLP-1 therapy has now produced statistically significant all-cause mortality reductions in two settings: FLOW (HR 0.80; 95% CI 0.67–0.95) in type 2 diabetes with CKD, and a pooled analysis of SELECT, FLOW, STEP-HFpEF, and STEP-HFpEF DM showing a roughly 19% lower mortality with semaglutide versus placebo. SUSTAIN-6 and LEADER each showed numerical mortality reductions that did not reach statistical significance individually but fit the same direction. The cumulative implication, drawn from more than 50,000 randomized participants, is that GLP-1 therapy reduces the risk of dying earlier from any cause in the populations studied. That is a claim very few medications have ever supported in randomized data.
How to read these numbers
Trial averages mask wide individual variation. Roughly one in seven STEP-1 participants lost less than 5% of body weight on semaglutide, and one in five SURMOUNT-1 participants on 15 mg tirzepatide lost less than 10%. [1][2] At the other tail, about 32% of SURMOUNT-1 completers on 15 mg lost more than 25% of baseline weight, and the top decile lost more than 30%. Response varies by genetics, baseline insulin resistance, sleep, stress, alcohol intake, and what else is on the plate; trial mean values describe a distribution, not a destiny.
Cardiovascular and kidney benefits in SELECT and FLOW were consistent across age, sex, BMI, and baseline-risk subgroups, but absolute risk reduction depends heavily on baseline risk. A patient with an established MI history gains far more in absolute terms than one with isolated risk factors. The number-needed-to-treat for one MACE prevented over five years was about 18 in SELECT for the highest-risk tertile and roughly 35 in the lowest. For kidney benefit in FLOW, the NNT was around 20.
Decisions about starting, switching, or stopping a GLP-1 should weigh expected benefits against tolerability, cost, supply, and the realistic plan for staying on therapy long term. The 16-week response checkpoint is a useful tool: patients who lose at least 5% of body weight by then are very likely to continue accumulating benefit, while those who do not should consider dose escalation, switching agent, or revisiting whether the medication is the right fit. The 12-month checkpoint is when the cardiovascular signals begin to compound; the 24-month checkpoint is where durability and lifestyle integration become decisive.
Across this evidence base, GLP-1 therapy has gone from a niche diabetes option to one of the most consequential drug classes of the last two decades, with documented benefits that now reach the heart, kidneys, liver, lungs, joints, brain reward circuits, and reproductive system. Phase 3 retatrutide trials, oral GLP-1 reformulations, and combination peptides under development suggest the next five years will further expand both the magnitude of weight loss and the breadth of conditions where these drugs deliver outcomes that older therapies could not match.
References
- Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1). N Engl J Med 2021;384:989-1002. doi:10.1056/NEJMoa2032183
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1). N Engl J Med 2022;387:205-216. doi:10.1056/NEJMoa2206038
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (SUSTAIN-6). N Engl J Med 2016;375:1834-1844. doi:10.1056/NEJMoa1607141
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes (SELECT). N Engl J Med 2023;389:2221-2232. doi:10.1056/NEJMoa2307563
- Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes (FLOW). N Engl J Med 2024;391:109-121. doi:10.1056/NEJMoa2403347
- Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the Treatment of Obstructive Sleep Apnea and Obesity (SURMOUNT-OSA). N Engl J Med 2024;391:1193-1205. doi:10.1056/NEJMoa2404881
- Aronne LJ, Horn DB, le Roux CW, et al. Tirzepatide as Compared with Semaglutide for the Treatment of Obesity (SURMOUNT-5). N Engl J Med 2025;393:26-36. doi:10.1056/NEJMoa2416394
- Knop FK, Aroda VR, do Vale RD, et al. Oral semaglutide 25 mg taken once daily in adults with overweight or obesity (OASIS-4). Lancet 2023;402:705-719.
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity: A Phase 2 Trial. N Engl J Med 2023;389:514-526. doi:10.1056/NEJMoa2301972
- Garvey WT, Batterham RL, Bhatta M, et al. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med 2022;28:2083-2091. doi:10.1038/s41591-022-02026-4
- Wilding JPH, Batterham RL, Davies M, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab 2022;24:1553-1564.
- Sorli C, Harashima SI, Tsoukas GM, et al. Efficacy and safety of once-weekly semaglutide monotherapy versus placebo in patients with type 2 diabetes (SUSTAIN 1). Lancet Diabetes Endocrinol 2017;5:251-260.
- Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes (SURPASS-2). N Engl J Med 2021;385:503-515. doi:10.1056/NEJMoa2107519
- American Diabetes Association. Standards of Care in Diabetes 2024: Pharmacologic Approaches to Glycemic Treatment. Diabetes Care 2024;47(Suppl 1):S145-S157.
- Marso SP, Bain SC, Consoli A, et al. SUSTAIN-6 (referenced above for primary CV outcome). N Engl J Med 2016;375:1834-1844.
- Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes (LEADER). N Engl J Med 2016;375:311-322. doi:10.1056/NEJMoa1603827
- Gerstein HC, Colhoun HM, Dagenais GR, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 2019;394:121-130.
- Husain M, Birkenfeld AL, Donsmark M, et al. Oral Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (PIONEER 6). N Engl J Med 2019;381:841-851. doi:10.1056/NEJMoa1901118
- McGuire DK, Marx N, Mulvagh SL, et al. Oral Semaglutide and Cardiovascular Outcomes in High-Risk Type 2 Diabetes (SOUL). N Engl J Med 2025;392:2001-2012. doi:10.1056/NEJMoa2501006
- Kosiborod MN, Abildstrøm SZ, Borlaug BA, et al. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity (STEP-HFpEF). N Engl J Med 2023;389:1069-1084. doi:10.1056/NEJMoa2306963
- Packer M, Zile MR, Kramer CM, et al. Tirzepatide for Heart Failure with Preserved Ejection Fraction and Obesity (SUMMIT). N Engl J Med 2025;392:427-437. doi:10.1056/NEJMoa2410027
- Sanyal AJ, Newsome PN, Kliers I, et al. Phase 3 Trial of Semaglutide in Metabolic Dysfunction-Associated Steatohepatitis (ESSENCE). N Engl J Med 2025;392:2089-2099.
- Loomba R, Hartman ML, Lawitz EJ, et al. Tirzepatide for Metabolic Dysfunction-Associated Steatohepatitis with Liver Fibrosis (SYNERGY-NASH). N Engl J Med 2024;391:299-310. doi:10.1056/NEJMoa2401943
- Bliddal H, Bays H, Czernichow S, et al. Once-Weekly Semaglutide in Persons with Obesity and Knee Osteoarthritis (STEP 9). N Engl J Med 2024;391:1573-1583. doi:10.1056/NEJMoa2403664
- Quddos F, Hubshman Z, Tegge A, et al. Semaglutide and tirzepatide reduce alcohol consumption in individuals with obesity. Sci Rep 2023;13:20998.
- Cummings JL, Atri A, Feldman HH, et al. evoke and evoke+: design of two large-scale, double-blind randomized phase 3 trials of oral semaglutide in early Alzheimer disease. Alzheimers Res Ther 2025;17:14.
- Salamun V, Jensterle M, Janez A, Vrtacnik Bokal E. Liraglutide increases IVF pregnancy rates in obese PCOS women with poor response to first-line reproductive treatments: a pilot RCT. Eur J Endocrinol 2018;179:1-11.
- Conte C, Hall KD, Klein S. Is Weight Loss-Induced Muscle Mass Loss Clinically Relevant? JAMA 2024;332:9-10. (See also lean-mass discussions in STEP-1 DXA substudies.)
- Kushner RF, Calanna S, Davies M, et al. Semaglutide 2.4 mg for the treatment of obesity: STEP trial patient-reported outcomes. Obesity (Silver Spring) 2020;28:1050-1061.
- Cleveland Clinic. GLP-1 Agonists. my.clevelandclinic.org/health/treatments/13901-glp-1-agonists