Compounded vs Brand-Name GLP-1
Summary: Compounded semaglutide and tirzepatide are not FDA-approved products and the regulatory window that allowed mass compounding closed in 2025, with the FDA moving in May 2026 to permanently exclude both molecules from the 503B bulk substances list. A narrower legal path remains under section 503A patient-specific compounding when a prescriber documents a clinical difference, and the brand pens are the safer default when accessible. A smaller, well-vetted set of licensed pharmacies still serves patients with documented clinical need at roughly one-third to one-fifth of cash brand pricing, while Lilly and Novo have aggressively cut their direct cash-pay prices in response. [1][2][3]
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.
What "compounded" actually means under US pharmacy law
Drug compounding is the practice of a licensed pharmacist combining or altering ingredients to make a medication for a specific patient. Two sections of the Federal Food, Drug, and Cosmetic Act define who can do this and how. Section 503A covers traditional pharmacies that prepare medications pursuant to a prescription for an identified individual patient. Section 503B covers outsourcing facilities that can produce larger batches without patient-specific prescriptions but must register with the FDA and follow current Good Manufacturing Practice (cGMP). State boards of pharmacy regulate 503A pharmacies; the FDA directly inspects 503Bs. [4][5]
Compounded drugs are not FDA-approved. The agency does not review them for safety, effectiveness, or quality before they reach patients. That is a deliberate trade off in the law: compounding exists so a clinician can address a patient need a manufactured product cannot, like a different strength, an excipient-free version for an allergy, or a dosage form a patient can actually use. It is not designed to be a parallel manufacturing channel for cheaper copies of approved drugs. [4]
The modern legal framework dates to the 2013 Drug Quality and Security Act (DQSA), which Congress passed in direct response to the New England Compounding Center (NECC) tragedy of 2012. NECC distributed contaminated injectable steroids that killed 64 patients and infected more than 750 across 20 states. Title I of the DQSA, the Compounding Quality Act, codified the 503A and 503B distinction and established outsourcing facilities as a regulated middle path between traditional pharmacy and full FDA-approved manufacturing. [6]
A 503A pharmacy operates under state board oversight, fills prescriptions for individually named patients, and is generally exempt from new-drug approval and cGMP requirements as long as it stays inside narrow statutory boundaries. A 503B outsourcing facility voluntarily registers with the FDA, undergoes federal inspection, follows cGMP, and may compound without a prescription for an identified patient, but its compounding is restricted to bulk substances on an FDA-published list (the 503B Bulks List) or to drugs in a current shortage. Both categories are forbidden from compounding products that are "essentially copies" of a commercially available, FDA-approved drug. [4][5]
The shortage rule and what changed in 2024-2025
Federal law generally bars compounding pharmacies from making copies of commercially available, FDA-approved drugs. The exception is when the agency has placed the drug on its official shortage list. Tirzepatide (Mounjaro and Zepbound) and semaglutide (Ozempic, Wegovy, Rybelsus) were both listed during 2022-2024, which opened the door for hundreds of pharmacies and outsourcing facilities to ship compounded versions through telehealth platforms. Compounded versions captured roughly 30 percent of US GLP-1 supply at the 2024 peak. [1][3]
Tirzepatide came off the FDA shortage list on October 2, 2024, was briefly reinstated, then declared resolved on December 19, 2024. Semaglutide was removed on February 21, 2025. The FDA set staggered cutoffs after each removal. 503A pharmacies had to stop compounding tirzepatide essentially immediately and stop compounding semaglutide by April 22, 2025. 503B outsourcing facilities had until March 19, 2025 for tirzepatide and May 22, 2025 for semaglutide. [7][8]
The Outsourcing Facilities Association (OFA) sued in the Northern District of Texas to keep the shortage status in place. Judge Mark T. Pittman upheld the FDA's authority on the tirzepatide case in March 2025 and a separate semaglutide challenge followed the same path. Eli Lilly and Novo Nordisk also pursued direct litigation against telehealth sellers and pharmacies, with Lilly filing suit against Mochi Health and several others in April 2025 and Novo expanding its suits through 2025 and into 2026. [7][9][10]
The 2026 turning point: FDA closes the bulks door
For most of 2025 the legal question was whether a 503A pharmacy could still compound semaglutide or tirzepatide for individual patients with a documented clinical difference, and whether a 503B outsourcing facility could compound from bulk under either the shortage exception or by petitioning to add the substance to the 503B Bulks List under a "clinical need" theory. The FDA answered both questions decisively in spring 2026.
On April 1, 2026, the agency issued a clarification reaffirming the "essentially copies" rule for 503A pharmacies and confirming that a compounded GLP-1 falls inside that prohibition when it shares the same active pharmaceutical ingredient (API) and route of administration as an approved product, even at slightly different strengths or with cosmetic add-ins. A safe-harbor provision allows a 503A pharmacy to dispense up to four prescriptions per month of an essentially-copies compound without enforcement risk, but volume above that threshold needs documented clinical justification per patient. [2]
Then on April 30, 2026, the FDA published a Federal Register proposal (docket 2026-08552) to formally exclude semaglutide, tirzepatide, and liraglutide from the 503B Bulks List. The agency rejected the argument that consumer affordability counts as the kind of "clinical need" the statute contemplates, stating in effect that pricing access is a payer, employer, manufacturer, and Medicare problem, not an FDA problem solved through bulk compounding. The Partnership for Safe Medicines and major brand manufacturers backed the move; outsourcing facilities and telehealth platforms opposed it. [1][3]
The combined effect: 503B outsourcing facilities lose the only remaining legal path to compound these molecules at industrial scale, and 503A pharmacies are pushed back inside the narrow patient-specific clinical-difference exception that the statute always intended. By April 2026 several major outsourcing facilities (Medisource, ProRx, BPI Labs) had already exited the GLP-1 compounding market in anticipation. [3]
Is compounded semaglutide legal right now?
Mass-market compounding of identical copies is no longer permitted now that the shortages are resolved and the FDA has signaled it will close the 503B bulk pathway as well. What 503A pharmacies can still do, with a valid prescription, is produce a compounded GLP-1 when a prescriber documents a clinical difference for a specific patient that the manufactured product cannot meet. In practice that means things like a non-standard titration dose between the manufacturer's strengths, a formulation without a particular excipient the patient reacts to, or combining the GLP-1 with another active ingredient where the prescriber judges it appropriate. [4][5]
This is a much smaller and narrower channel than the 2023-2024 boom. The FDA has signaled it will scrutinize whether "clinical difference" claims are real or pretextual, and on March 3, 2026 the agency publicly released 30 warning letters to telehealth and online wellness clinics for false or misleading marketing of compounded GLP-1s, including claims that compounded products contained "the same ingredients" as branded versions or that they had been clinically tested. Recipients had 15 working days to respond, with seizure and injunction on the table for non-compliance. [11]
State pharmacy boards add another layer. Oregon, Ohio, and Alabama have all issued enforcement-oriented position statements on compounded semaglutide, with some banning specific salt forms outright and others restricting which APIs and excipients are allowed. Med spas have come under particular scrutiny: only 13 states have any guidance covering med spas, and only Ohio requires med spas administering compounded GLP-1s to hold a pharmacy board license. [12][13]
What "clinical difference" actually means
The clinical-difference exception is the single most important concept for understanding what a 503A pharmacy can legitimately compound after April 2025. The statute requires a prescriber to identify a "change that produces a significant difference for an identified individual patient" between the compounded product and the FDA-approved drug. The FDA has provided guidance on what does and does not qualify. [14]
Genuine clinical differences include:
- Documented allergy to an excipient in the brand pen (for example, a patient with a known reaction to a specific preservative or buffer).
- Inability to use the device. Patients with arthritis, vision impairment, or cognitive issues who cannot operate the manufacturer's pen reliably may need a vial-and-syringe presentation a clinician supervises.
- Pediatric or weight-band dosing that falls between the manufacturer's titration steps and is not commercially available.
- Slow-titration micro-dosing for patients who could not tolerate the standard titration schedule and where the prescriber documents the clinical reason.
Combinations of semaglutide with vitamin B12 (cyanocobalamin), glycine, levocarnitine, or similar add-ins were heavily marketed during the shortage as "clinical differences" justifying compounding. The clinical evidence behind the combinations is thin. Semaglutide has not been studied in combination with B12, and supplementing B12 in patients with normal B12 status confers no documented benefit. The FDA's April 2026 guidance specifically called out semaglutide-plus-B12 as an example of a product that is still considered an essential copy when strengths and route of administration are within roughly 10 percent of the commercial equivalent. [2][15]
That does not mean every combination compound is illegitimate. It does mean a prescriber needs an actual clinical reason on the chart, not a marketing claim, and that pharmacies relying on combination products as a workaround are on increasingly thin legal ice.
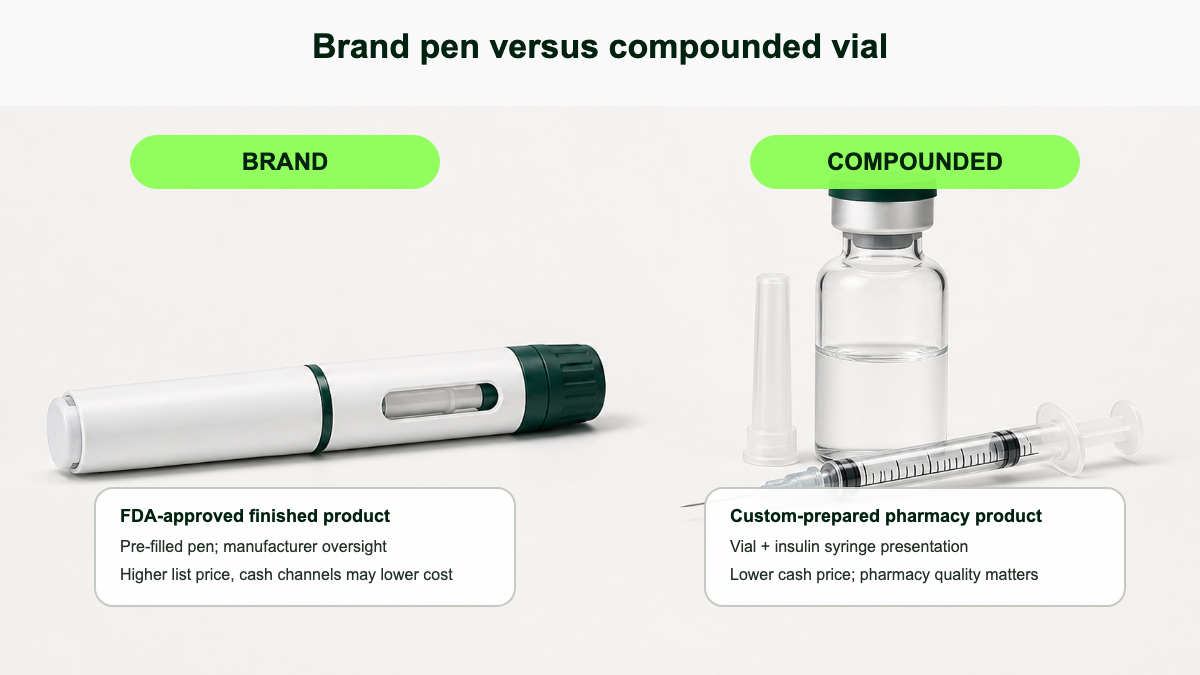
How the molecule compares: brand vs compounded
When a 503A or 503B pharmacy uses pharmaceutical-grade semaglutide or tirzepatide active pharmaceutical ingredient (API) sourced from an FDA-registered facility, the active molecule is the same one in the brand product. The differences are in everything around the molecule: the inactive ingredients, the buffer system, the concentration, the container, and the quality testing behind the batch. [16][17]
A real concern the FDA has flagged is salt forms. Some products marketed as "semaglutide" have actually contained semaglutide sodium or semaglutide acetate, which are different chemical entities than the semaglutide base used in Ozempic, Wegovy, and Rybelsus. The agency has stated there is no evidence those salt forms are safe or effective, and they are not the molecule that was studied in the clinical trials supporting approval. Novo Nordisk does not sell semaglutide API to compounders, so any compounded "semaglutide" product is sourced from a third-party API manufacturer (overwhelmingly in China and India), and a 2024 Brookings analysis found that nearly 25 percent of the semaglutide-shipping facilities surveyed had never been inspected by the FDA, with three Chinese firms responsible for 44.5 percent of imported volume cited for cGMP violations. [16][18]
| Attribute | Brand (Ozempic, Wegovy, Mounjaro, Zepbound) | Compounded (legitimate 503A pharmacy) |
|---|---|---|
| FDA approval | Approved drug product with NDA | Not FDA-approved as a product |
| Active ingredient | Semaglutide or tirzepatide base | Should be the same base; salt forms are a red flag |
| Manufacturing oversight | FDA cGMP at brand facilities | State board for 503A; FDA cGMP for 503B outsourcing facilities |
| API source | Brand-controlled, fully validated supply chain | Third-party API manufacturer, often overseas; quality varies |
| Presentation | Prefilled multi-dose pen, fixed dose increments | Typically vial plus insulin syringe or prefilled syringe; patient measures units |
| Dose flexibility | Manufacturer titration steps only | Custom microdoses possible (clinically justified) |
| Insurance coverage | Possible with prior authorization | Almost always cash-pay |
| Typical monthly cost (cash) | $349-$499 self-pay direct; up to $1,000-$1,350 list | $150-$400 for semaglutide; $300-$600 for tirzepatide |
| Clinical trial data | Backed by STEP, SUSTAIN, SURMOUNT, SURPASS programs | No product-specific trials; relies on the brand evidence base |
| Adverse event reporting | Mandatory MedWatch and post-market surveillance | Variable; 503Bs report, 503As often do not at scale |
| Legal status (May 2026) | Unrestricted prescribing | 503A only with documented clinical difference; 503B path closing |
Why compounded is cheaper, and what the price gap actually represents
The brand price reflects the cost of the original clinical development, ongoing safety surveillance, manufacturer margins, supply chain, and pharmacy benefit manager economics. Novo Nordisk's published US list prices for Ozempic and Wegovy historically sat above $1,000 per month before rebates, and Lilly's Mounjaro and Zepbound were in a similar range. Both companies launched and then aggressively cut direct cash-pay channels in 2024-2026 (LillyDirect and NovoCare) in direct response to compounded competition. [1][19]
By December 2025, Lilly had cut LillyDirect single-dose Zepbound vial pricing again, with the lowest doses landing under $300 per month for cash patients enrolled in the self-pay journey program. Novo followed with comparable Wegovy pen pricing through NovoCare around the same window. The result is that the brand-vs-compounded math, which used to be roughly $1,300 vs $250, is now closer to $349-$499 vs $150-$400 for many patients. The gap is still real, but it is no longer a 5x difference for everyone. [19][20]
Compounded versions skip the new-drug development cost and most marketing, source API from third-party producers, and ship in vials rather than complex pen devices. Cash prices through telehealth platforms have generally landed in the $150-$400 range for semaglutide and $300-$600 for tirzepatide. The gap is real, but so is what the gap pays for: a regulated supply chain with inspected facilities, batch records, an approved label, mandatory adverse event surveillance, and a device validated for the dose strengths shown in clinical trials.
Consumers also need to factor in what the headline price excludes. A telehealth platform's $199 monthly compounded fee may not include the initial clinician consultation, lab work where appropriate, refrigerated shipping, syringes and sharps disposal, follow-up visits, or ongoing prescription renewals. Brand cash programs often bundle prescription, shipping, and pharmacy support. The honest comparison is total annual out-of-pocket, not the lowest sticker number on the landing page.
Quality concerns to take seriously
The FDA has received hundreds of adverse event reports tied to compounded semaglutide and tirzepatide. As of late 2024, at least 10 deaths and more than 100 hospitalizations had been associated with compounded semaglutide in FDA adverse event reports, though the agency notes those reports are unverified and do not establish causation. The dominant pattern is dosing errors traced to patients drawing the wrong number of units from vials, plus a smaller share of infection and quality complaints from non-sterile or contaminated products. Common contributing factors include unfamiliar concentrations, instructions in "units" that do not map cleanly to milligrams, and vial labeling that differs between pharmacies. [16][21]
Beyond dosing errors, four quality questions matter:
- API source. The active ingredient should come from a facility registered with the FDA, ideally listed on a Drug Master File (DMF), and inspected for compliance with USP standards. Pharmacies should be able to name the supplier and provide a certificate of analysis (CoA) on request. The Brookings analysis of import records found that one reviewed CoA allowed up to 15 percent non-semaglutide content as acceptable; a real CoA from a credible API maker should show much tighter purity specifications. [18]
- Sterility, potency, and endotoxin testing. Sterile injectables require third-party testing for sterility (USP <71>), endotoxins (USP <85>), potency, and stability. Reputable 503A pharmacies engage independent FDA-registered analytical labs and can produce batch-specific test results.
- Salt form and synthetic peptide quality. The label should say semaglutide or tirzepatide, not semaglutide sodium or semaglutide acetate. USP general chapters <1503> (Quality Attributes of Synthetic Peptide Drug Substances) and <1504> (Quality Attributes of Starting Materials for Chemical Synthesis) describe the expected purity profile. Anything sold as "research only" or "not for human use" is exactly that, and using it as a medication is unsafe and unlawful. [16][22]
- NABP and state board status. The National Association of Boards of Pharmacy (NABP) tracks online pharmacies and publishes a rolling list of "not recommended" sites. NABP analysis through January 2025 found that nearly 200 GLP-1 bulk shipments entered the US despite "clear legal issues," many through de minimis (under $800) packages that bypass routine customs inspection. Any pharmacy you cannot verify on your state board's license-lookup tool is a hard pass. [13]
Red flags and green flags when evaluating a provider
The legitimate end of this market and the dangerous end can look similar in a Google ad. The differences show up in the details.
Red flags:
- No named, licensed pharmacy you can look up on your state board's website
- No clinician evaluation, or a "questionnaire only" model with no real prescriber review or follow-up
- Same dose for everyone regardless of weight, history, or response
- Sales of "research peptides," vials labeled "not for human use," or unbranded packaging
- No certificate of analysis available for the batch
- Labels that read "semaglutide sodium" or "semaglutide acetate" rather than semaglutide [16]
- Marketing that calls the product "the same as Ozempic" or "FDA-approved semaglutide" (both are misrepresentations the FDA explicitly cited in its 2026 warning letters) [11]
- Prices below roughly $100 per month for semaglutide or $150 for tirzepatide, which generally cannot cover real API, testing, sterile fill, and clinical oversight
- Cryptocurrency-only or wire-transfer-only payment, anonymous corporate ownership, no US street address
- International sourcing without verified US import records or registered foreign establishment
- No process for follow-up, side effect management, or stopping the medication
Green flags:
- Named 503A pharmacy in good standing with the state board, or a registered 503B with FDA inspection history you can verify on the FDA website
- Board-certified clinician (MD, DO, NP, or PA) reviewing your medical history before prescribing, with an active license in your state
- Dose tied to your starting weight, comorbidities, and titration response
- Documented clinical reason on file for using a compounded rather than brand product, consistent with the FDA clinical-difference standard
- API sourced from an FDA-registered facility on a US Drug Master File, with a real CoA per batch
- Independent third-party potency, sterility, and endotoxin testing
- Glass vials with batch and lot numbers, refrigerated shipping with temperature monitoring
- HIPAA-compliant intake and messaging platform
- Clear labeling, written instructions in milligrams and units, and a way to reach a pharmacist or clinician between visits
- A plan for monitoring, side effects, and transitioning to brand if appropriate
Litigation timeline and major cases
The compounded GLP-1 dispute has produced one of the most active pharmaceutical litigation dockets of the past three years.
- October 2024 - March 2025: OFA v. FDA (tirzepatide). The Outsourcing Facilities Association sued in the Northern District of Texas to keep tirzepatide on the shortage list. Judge Mark T. Pittman denied the preliminary injunction in March 2025, upholding the FDA's decision. [9]
- April 23, 2025: Lilly v. compounders. Lilly filed simultaneous lawsuits against Mochi Health, Fella Health, Willow Health, and Henry Meds for selling tirzepatide-niacinamide and similar combination compounds the company called "deceptive" copies of Zepbound. [10]
- February 9, 2026: Novo Nordisk v. Hims & Hers. Novo filed federal patent infringement litigation against Hims for marketing compounded semaglutide and oral semaglutide formulations. [23]
- March 3, 2026: FDA warning letters. The FDA released 30 warning letters to telehealth and wellness clinics for false or misleading compounded GLP-1 marketing under FDCA Sections 502(a) and 502(bb). [11]
- March 9, 2026: Hims-Novo settlement. Novo dropped the lawsuit (with right to refile) in exchange for Hims discontinuing compounded GLP-1 advertising and offering branded Wegovy and oral semaglutide on its site. Hims retained the ability to dispense compounded GLP-1s only when a provider determines clinical necessity. [24]
- April 1, 2026: FDA "essentially copies" clarification. The agency reaffirmed that compounded GLP-1s sharing the same API and route as a branded product are essential copies, with a four-prescription-per-month safe harbor for 503A pharmacies. [2]
- April 30, 2026: Federal Register 503B bulks proposal. The FDA proposed permanently excluding semaglutide, tirzepatide, and liraglutide from the 503B Bulks List. [3]
Several patterns are clear from the litigation. Brand manufacturers have shifted strategy from purely targeting compounders to targeting telehealth platforms that market compounded products, since the platforms are the consumer-facing brand. Settlements typically include the platform pivoting to a brand-distribution arrangement rather than a cash payment. And courts have consistently sided with the FDA's authority to declare shortages resolved.
Manufacturer counter-pricing and why it exists
One of the more interesting market dynamics of 2024-2026 is that brand manufacturer cash-pay pricing has fallen by roughly 50 to 70 percent in just two years, almost entirely because of compounding pressure. LillyDirect launched in early 2024 with $549 vials of Zepbound. By December 2025 the lowest dose landed near $299 cash for self-pay journey enrollees. NovoCare introduced direct cash-pay Wegovy pens at $499 monthly in 2025 and continued to expand discount tiers through 2026. [19][25]
That this happened so quickly is unusual in pharmaceutical pricing and was not driven by patent expiry, generic competition, payer pushback, or regulatory mandate. It was driven by compounded competition. The brand companies do not advertise this connection, but the timing is unmistakable: every major LillyDirect and NovoCare price cut tracks a compounding-related event. The practical implication for patients is that shopping the brand cash-pay channels first is now a much better deal than it was a year ago, and the gap between brand cash and compounded cash is narrower than it has ever been.
This dynamic also helps explain why Lilly and Novo have pursued litigation as aggressively as they have. Compounded competition is the single most effective price-discipline force the GLP-1 market has experienced. Closing the compounded channel restores pricing power for brands; the cash-pay programs they rolled out as a response will most likely loosen once compounded competition disappears.
Patent expiration and the post-2031 landscape
The other major variable is patent expiration. Novo Nordisk's primary US composition-of-matter patent on semaglutide ('343 patent) is set to expire in December 2031, extended from the original ~2025 date through Patent Term Extension. After settling with Mylan, Dr. Reddy's, Apotex, and Sun Pharmaceuticals in late 2024, Novo is positioned to face generic Ozempic entry in approximately 2032. Wegovy and Rybelsus have additional formulation and method-of-use patents that extend into the late 2030s. [26]
Lilly's tirzepatide composition patents extend further, with US generic competition unlikely before the mid-2030s. Internationally the timeline is faster: semaglutide patents expire in India, Canada, China, Brazil, and Turkey starting in 2026, and over 17 manufacturers in China and 10 in India have advanced semaglutide candidates in clinical trials, with prices in those markets expected to fall by 50 to 75 percent. [27]
For US patients the practical reading is that compounded GLP-1s were a temporary affordability bridge that the FDA is now closing, and approved generic semaglutide at the lowest cash prices is still 5 to 7 years away. In between, the brand cash-pay programs and a much-narrower clinical-difference compounding channel are the realistic options.
Decision framework: should you consider compounded?
The honest framing: if you can access an FDA-approved brand pen at a price you can sustain, that is the lower-risk default. The clinical evidence base, the device, and the regulatory oversight are all built around the brand product. Lilly and Novo's direct cash programs have meaningfully narrowed the affordability gap for many patients. [1][19]
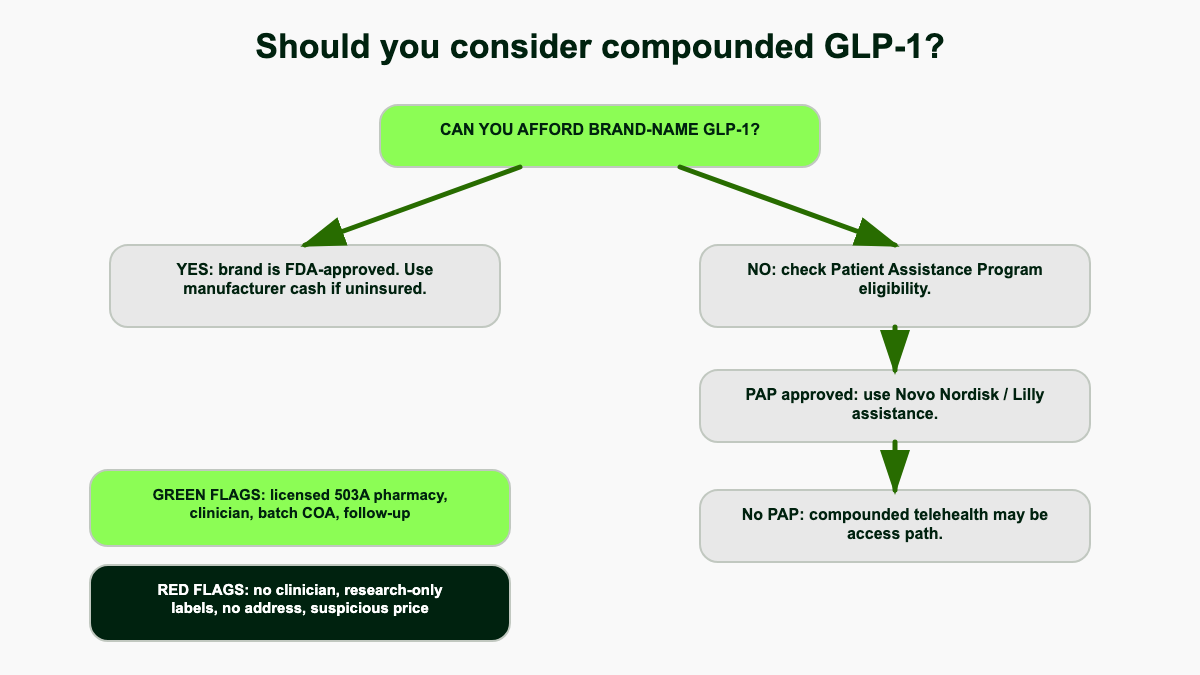
A compounded GLP-1 may still make sense if:
- You have a documented clinical need a manufactured product cannot meet (genuine excipient allergy, inability to use the device, a non-standard dose with a real clinical reason).
- You are working with a licensed 503A pharmacy in good standing and an evaluating clinician licensed in your state.
- You understand you are taking a product that has not been individually reviewed by the FDA.
- Brand cash pricing through LillyDirect or NovoCare is genuinely unaffordable in your situation, and you have explored manufacturer patient assistance programs.
It rarely makes sense if your only reason is convenience, the cheapest possible price from an unfamiliar website, or an inability to wait for a brand prescription to be processed.
Patients who should be especially cautious about compounded products include people who are pregnant or trying to conceive, people with a personal or family history of medullary thyroid carcinoma or MEN 2, people with a history of pancreatitis, and anyone managing a complex medication regimen where dosing precision matters. The FDA's standing recommendation is that patients use approved products when they are available. [16]
What to ask before you commit
Whether you are evaluating a brand cash-pay program or a compounded option, the following questions will surface most of the information that matters.
- Is the pharmacy named, US-based, and listed in good standing on its state board's online roster?
- If the pharmacy is a 503B outsourcing facility, when was its last FDA inspection and what did the inspection find?
- Who is the prescribing clinician and what is their license number? Can you verify it on the state medical or nursing board?
- Does the prescriber actually review your full medical history, including comorbidities, family history of thyroid cancer, gallbladder disease, pancreatitis, current medications, and pregnancy plans?
- If compounded, what specific clinical-difference reason does the prescriber document for using a compounded versus a branded product?
- Where does the API come from, and is the manufacturer FDA-registered with a US Drug Master File on file?
- Does each batch come with a certificate of analysis showing potency, purity, sterility, and endotoxin results?
- What is the salt form? Anything other than semaglutide base or tirzepatide base is a refusal point.
- What is the total annual cost including consultation, lab work, shipping, and follow-up visits, not just the monthly headline price?
- What happens if you have a side effect, run out of medication while traveling, or want to switch to brand? Who do you call?
The telehealth platform landscape
The cash-pay telehealth segment that grew up around compounded GLP-1s is in active reorganization. Understanding which platforms do what helps consumers triangulate where the market is moving.
- Hims & Hers. Sold compounded semaglutide aggressively from mid-2024, was sued by Novo Nordisk in February 2026, settled in March 2026, and now offers branded Wegovy and oral semaglutide alongside a residual compounded program limited to clinically necessary cases. The settlement is widely viewed as a template for how other large platforms will resolve manufacturer disputes. [24]
- Ro (formerly Roman). Maintains a hybrid program with both branded GLP-1 access and a clinically gated compounded option through partner pharmacies. Has not been a defendant in major litigation through May 2026.
- Sequence (Weight Watchers). Pivoted toward branded GLP-1 dispensing earlier than most platforms, in part because of WW's broader regulatory exposure on weight loss claims. Not a major compounded supplier in 2026.
- Henry Meds, Mochi Health, Fella Health, Willow Health. All four named in Lilly's April 2025 tirzepatide-niacinamide lawsuits. As of May 2026 several have scaled back compounded tirzepatide while maintaining compounded semaglutide programs that rely on the clinical-difference exception. [10]
- Found, Calibrate, Noom Med, Eden, ReflexMD, Alan Meds, GetThinMD. Smaller players with varying degrees of compounded inventory and varying quality of clinician oversight. The Wired investigative series in 2024 documented wide variation across this group on questionnaire rigor and clinical screening. [30]
- Independent 503A pharmacies. A long tail of regional 503A pharmacies operate compounded GLP-1 programs through individual prescribers. Quality varies dramatically; the state board lookup is the critical filter.
The general direction across the market is consolidation around branded distribution partnerships, with compounded inventory increasingly reserved for documented clinical-difference cases. Platforms that built their model on undifferentiated compounded copies are facing the most legal pressure and the most pricing pressure from manufacturer cash-pay programs simultaneously.
How brand manufacturers' counter-narrative differs from the FDA's
It is worth separating Novo Nordisk and Lilly's public statements on compounded GLP-1s from the FDA's actual regulatory position, because they overlap but are not identical.
The brand companies frame compounded GLP-1s as fundamentally counterfeit or unsafe regardless of source, repeatedly emphasizing the deaths and adverse events linked to compounded versions and pointing to international API sourcing as inherently risky. Novo CEO Lars Fruergaard Jorgensen's "there is only one semaglutide" framing leaves little room for the legitimacy of any compounded product. [21]
The FDA's position is more nuanced. The agency consistently states that compounded drugs are not FDA-approved and that patients should use approved products when accessible, but it also explicitly preserves the 503A clinical-difference compounding pathway and acknowledges legitimate pharmacy compounding as part of the statutory framework. FDA Commissioner Marty Makary's 2026 statements on the warning letters specifically called out misleading marketing rather than compounding itself. [11][16]
The honest read of both is that quality varies enormously across compounded providers, that a well-run 503A pharmacy with USP-grade API and full sterility testing produces something genuinely close to the brand product, and that an offshore "research peptide" repackager produces something genuinely dangerous, and that lumping all of these together as either "the same thing as Ozempic" or "counterfeit poison" both miss the actual situation. The patient's job is to figure out where any specific provider sits on that spectrum.
International and cross-border purchasing
Some patients have explored buying semaglutide or tirzepatide internationally, either because the price is lower in their home country or because they want to source the brand product directly from a foreign pharmacy. A few practical points worth knowing.
Importing prescription drugs into the US for personal use is generally illegal under the Federal Food, Drug, and Cosmetic Act, with a narrow personal-importation enforcement-discretion policy that has historically applied only when the patient has a serious condition, no domestic alternative, the quantity is limited (typically a 90-day supply), and the drug is for the patient's personal use. Compounded weight-loss medication does not fit any of those criteria. [16]
Counterfeit Ozempic pens have been documented in the US supply chain, with the FDA issuing recall and counterfeit alerts during the 2023-2024 shortage period when demand vastly outstripped legitimate supply. The most common counterfeit signal was packaging that looked authentic but contained the wrong dose strength, the wrong preservative system, or no active ingredient at all. The counterfeit risk has decreased as supply has stabilized but has not gone to zero, particularly for product purchased through unverified online channels.
For US patients the practical answer is that there is no safe, legal cross-border path to buying semaglutide or tirzepatide cheaply from abroad. The legitimate options remain US-licensed pharmacy with US-licensed prescriber, whether dispensing a brand pen or a compounded product within the 503A clinical-difference framework.
Where this is heading
The 2025 court rulings sided with the FDA's authority to declare shortages resolved, the brand manufacturers have continued to expand cash-pay direct channels, the FDA has moved to permanently close the 503B bulk pathway for semaglutide and tirzepatide, and the largest telehealth platforms have shifted toward partnerships with Lilly and Novo or have wound down their compounded books. A smaller, more clinically focused 503A compounding channel persists for patients with real individualized needs, and that channel is likely to be the long-term shape of the market. [1][3][24]
If you are evaluating options now, the practical sequence is: check your insurance coverage for the brand product, check the manufacturer direct programs (LillyDirect, NovoCare), explore manufacturer patient assistance and copay assistance programs, and only consider a compounded route through a licensed pharmacy and an evaluating clinician who can document why a compounded version is appropriate for your situation. Compounded GLP-1s solved a real affordability gap during the 2022-2025 shortage. The window has narrowed significantly and the legal ground continues to shift, but for the right patient with the right provider it remains a legitimate option in 2026.
References
- Fierce Healthcare. "FDA declares semaglutide shortage over, spelling end to compounded GLP-1 market." February 2025. fiercehealthcare.com
- Foley & Lardner LLP. "FDA Clarifies Policies for Pharmacy Compounders of GLP-1 Products." April 2026. foley.com
- Partnership for Safe Medicines. "FDA moves to restrict large-scale compounding of GLP-1s." May 4, 2026. safemedicines.org
- US Food and Drug Administration. "Compounding and the FDA: Questions and Answers." fda.gov
- US Food and Drug Administration. "Compounding Laws and Policies." fda.gov
- Wikipedia. "Drug Quality and Security Act." wikipedia.org
- Foley & Lardner LLP. "GLP-1 Drugs: FDA Removes Semaglutide From the Drug Shortage List." February 2025. foley.com
- Harris Beach Murtha. "GLP-1 Weight-Loss Drugs Off Shortage List: Deadlines to Stop Compounding." 2025. harrisbeachmurtha.com
- Foley & Lardner LLP. "Compounded GLP-1 Drugs: Texas Judge Denies PI Motion." March 2025. foley.com
- NPR. "Eli Lilly sues companies selling alternative versions of its weight loss drug." April 23, 2025. npr.org
- Frier Levitt. "FDA Warning Letters and the Hims-Novo Nordisk Deal." March 2026. frierlevitt.com
- Buchanan Ingersoll & Rooney. "State Board of Pharmacy: Ohio GLP-1 Compounders Be Vigilant." bipc.com
- National Association of Boards of Pharmacy. "Med Spas and GLP-1 Safety." nabp.pharmacy
- US Food and Drug Administration. "Compounded Drug Products That Are Essentially Copies of a Commercially Available Drug Product Under Section 503A." fda.gov
- SingleCare. "Compounded semaglutide with B12: Benefits and risks." singlecare.com
- US Food and Drug Administration. "FDA's Concerns With Unapproved GLP-1 Drugs Used for Weight Loss." fda.gov
- Healthline. "Is Compounded Semaglutide the Same as Ozempic?" 2025. healthline.com
- Brookings Institution. "The Wild East of semaglutide." brookings.edu
- Eli Lilly. "Lilly lowers the price of Zepbound single-dose vials." lilly.com
- Novo Nordisk. "Wegovy Online Pharmacy and Savings." novocare.com
- CNN. "Compounded semaglutide associated with at least 10 deaths." November 6, 2024. cnn.com
- US Pharmacopeia. "GLP1 Therapies Peptide Standards." usp.org
- Hall Render. "GLP-1 Gold Rush Meets Legal Reality: Novo Nordisk Challenges Compounded Alternatives." March 2026. hallrender.com
- STAT News. "Novo Nordisk, Hims reach deal to end bitter dispute over Wegovy." March 9, 2026. statnews.com
- CNBC. "Eli Lilly cuts cash prices of Zepbound weight loss drug vials." December 1, 2025. cnbc.com
- Markman Advisors. "What is the patent landscape for Novo Nordisk's semaglutide products: Ozempic, Wegovy and Rybelsus." February 2025. markmanadvisors.com
- IQVIA. "Off-patent semaglutide in 2026: the next revolution in anti-obesity treatment." July 2025. iqvia.com
- JAMA Health Forum. "Marketing and Safety Concerns for Compounded GLP-1 Receptor Agonists." January 2025. jamanetwork.com
- Holland & Knight. "Eli Lilly Strikes Back Against Pharmacy Compounders and Telehealth." June 2025. hklaw.com
- GoodRx. "Compounded Semaglutide: What Is It and Is It Safe?" 2025. goodrx.com
- Wired. "It's Shockingly Easy to Buy Off-Brand Ozempic Online." 2024. wired.com