Semaglutide (Ozempic, Wegovy)
Summary: Semaglutide is a once-weekly GLP-1 receptor agonist sold as Ozempic and Rybelsus for type 2 diabetes and as Wegovy for chronic weight management. STEP 1 trial participants on the 2.4 mg dose lost roughly 14.9% of body weight over 68 weeks [1], and the SELECT trial showed a 20% relative reduction in major cardiovascular events over a median 40 months [3].
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.
What semaglutide is
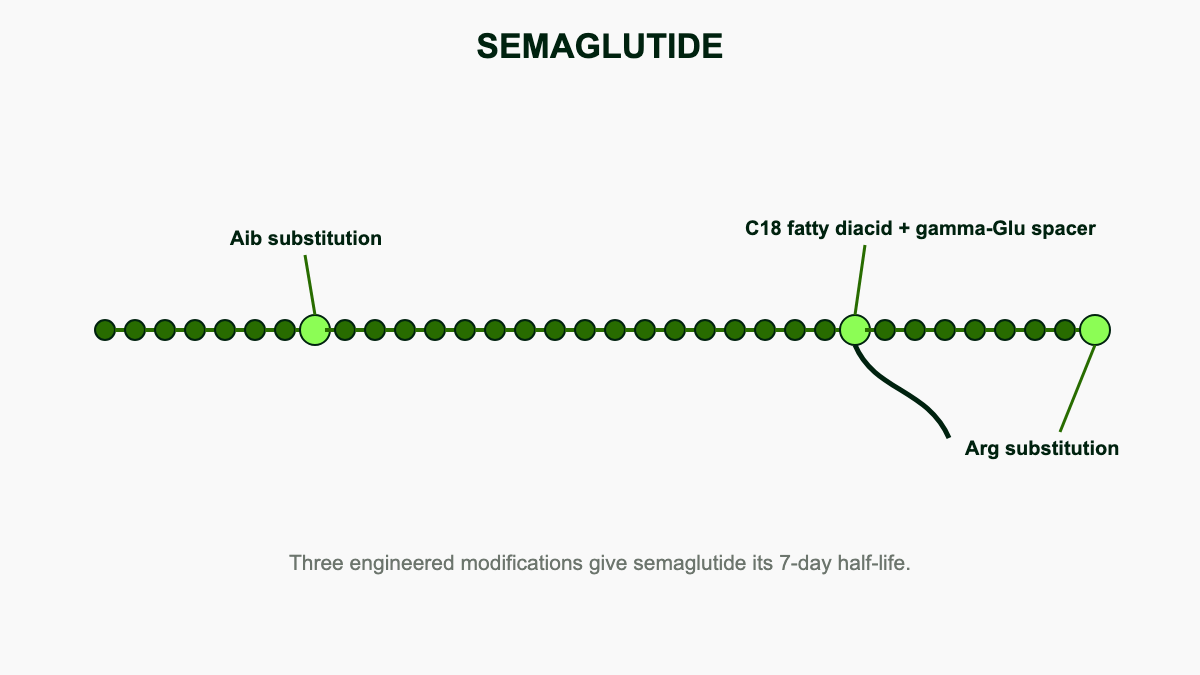
Semaglutide is a synthetic version of GLP-1, a gut hormone the body releases after eating. Novo Nordisk modified the natural peptide in two places: an alanine swap at position 8 to block degradation by the DPP-4 enzyme, and an 18-carbon fatty acid chain attached at lysine 26 so the molecule binds tightly to albumin in the bloodstream [10]. Albumin binding exceeds 99%, which protects the peptide from renal filtration and proteolytic breakdown and produces a terminal half-life of roughly 165 hours, or about seven days [11]. That long half-life is what makes once-weekly subcutaneous dosing possible.
The same molecule is sold under three brand names. Ozempic is the subcutaneous pen approved in December 2017 for type 2 diabetes [12]. Rybelsus is the oral tablet approved in September 2019, the first GLP-1 in pill form [13]. Wegovy is the higher-dose subcutaneous pen approved in June 2021 for chronic weight management [14]. A fourth product, Wegovy in tablet form at 25 mg, received FDA approval in late 2025 and reached pharmacies in early January 2026 [15]. All four are made by Novo Nordisk, which holds composition-of-matter patents through 2031 in the United States.
The development story traces back to liraglutide (Victoza, Saxenda), the once-daily GLP-1 analog approved in 2010. Novo Nordisk chemists, led by Lotte Bjerre Knudsen, replaced liraglutide's 16-carbon palmitoyl chain with an 18-carbon diacid linker and added a glutamate spacer, raising albumin affinity by an order of magnitude [10]. The result was the first GLP-1 receptor agonist that could be dosed weekly rather than daily and the first that could be packaged with a permeation enhancer for oral absorption.
How it works in the body
Semaglutide binds to GLP-1 receptors in the pancreas, brain, gut, and several other tissues. In the pancreas it triggers insulin release in a glucose-dependent way, meaning it only acts when blood sugar is elevated, which limits hypoglycemia risk [4]. It also suppresses glucagon, the hormone that tells the liver to dump stored glucose.
The weight effects come from a different set of receptors. In the hypothalamus, semaglutide reduces hunger signals and increases satiety [4]. In the stomach, it slows gastric emptying so meals stay in the stomach longer and people feel full sooner. The combined effect is fewer calories eaten without conscious restriction, which is why the food-noise reduction is the symptom most patients describe first. For deeper mechanism detail, see the dedicated page on how GLP-1 works.
The three brands at a glance
The three semaglutide products share an active ingredient but differ in dose ceiling, route, and approved indications. Ozempic is dosed weekly by injection at 0.25, 0.5, 1.0, or 2.0 mg and is approved for glycemic control, MACE reduction in type 2 diabetes with established heart disease, and slowing kidney disease progression [12]. Wegovy injection is dosed weekly at up to 2.4 mg and is approved for chronic weight management, MACE reduction in cardiovascular disease with overweight or obesity, and noncirrhotic MASH with stage 2-3 fibrosis [14][16]. Rybelsus is the once-daily oral tablet at 3, 7, or 14 mg, approved for glycemic control and, since October 2025, MACE reduction in adults with type 2 diabetes at high cardiovascular risk [13][17].

Brand pricing also differs sharply. Ozempic and Rybelsus carry list prices around $968 to $1,029 per month, while Wegovy lists at roughly $1,349 [18]. Self-pay through NovoCare Pharmacy lowers Wegovy's monthly cost to $499 for the 2.4 mg maintenance dose for cash-paying patients without insurance, and to $199 for the starter doses through June 2026 [19]. See cost and access for a full pricing breakdown.
FDA approval timeline
The FDA has expanded semaglutide's label more than ten times since 2017. The major milestones [12][13][14][15][16][17][20]:
- December 2017: Ozempic approved for glycemic control in type 2 diabetes
- September 2019: Rybelsus approved for glycemic control in type 2 diabetes (first oral GLP-1)
- January 2020: Ozempic adds MACE reduction in type 2 diabetes with established cardiovascular disease (based on SUSTAIN 6)
- June 2021: Wegovy approved for chronic weight management in adults with BMI 30+, or 27+ with a weight-related comorbidity
- December 2022: Wegovy expanded to adolescents 12 and older with obesity (based on STEP TEENS)
- March 2024: Wegovy adds MACE reduction in cardiovascular disease with overweight or obesity (based on SELECT)
- January 2025: Ozempic adds reduction of kidney disease progression in type 2 diabetes with chronic kidney disease (based on FLOW)
- August 2025: Wegovy approved for noncirrhotic MASH with moderate to advanced fibrosis (based on ESSENCE)
- October 2025: Rybelsus adds MACE reduction in type 2 diabetes at high cardiovascular risk (based on SOUL)
- December 2025: Oral Wegovy 25 mg approved for chronic weight management (first oral GLP-1 for obesity)
Behind that timeline sits roughly $5.7 billion in cumulative phase 3 spending from Novo Nordisk and the largest set of cardiovascular outcomes trials ever run for any single drug class. Ten major outcomes programs have read out so far: STEP, SUSTAIN, PIONEER, SELECT, FLOW, SOUL, STRIDE, STEP-HFpEF, ESSENCE, and OASIS.
Pharmacokinetics and pharmacodynamics
After subcutaneous injection, semaglutide reaches peak plasma concentration in 1 to 3 days [11]. The terminal half-life is roughly 165 hours, and steady state is achieved after 4 to 5 weeks of weekly dosing [11]. Apparent volume of distribution is about 12.5 L, consistent with confinement to the vascular compartment by extensive albumin binding. Clearance is roughly 0.05 L per hour. Elimination occurs through proteolytic degradation; only about 3% of an administered dose is excreted unchanged in urine, which is why no dose adjustment is required for any degree of renal or hepatic impairment [21].
Oral semaglutide (Rybelsus) achieves bioavailability of roughly 0.4 to 1% through co-formulation with sodium N-(8-[2-hydroxybenzoyl]amino) caprylate (SNAC), a permeation enhancer that transiently buffers gastric acid and opens tight junctions in the gastric epithelium [13][22]. Tmax for the oral form is about 1 hour. The terminal half-life still runs around 7 days because once absorbed, the molecule behaves identically to the injected form. Steady state with oral dosing also takes 4 to 5 weeks.
Pharmacodynamically, semaglutide produces a flat plasma profile across the dosing week, which contrasts with the peaks-and-troughs of daily exenatide. The flat profile is one reason gastrointestinal tolerability tends to be better than with shorter-acting GLP-1 analogs, though it also means side effects from a dose increase build slowly across the four weeks after each titration step.
Dosing and titration
Dose escalation is slow, deliberate, and the same regardless of brand goal: limit gastrointestinal side effects by giving the gut time to adapt.
Wegovy (subcutaneous, weight management)
Wegovy uses a five-step titration: 0.25 mg weekly for 4 weeks, then 0.5 mg, then 1.0 mg, then 1.7 mg, and finally 2.4 mg, each held for 4 weeks before stepping up [14]. Full titration takes 16 weeks, and 2.4 mg is the maintenance dose. If a patient cannot tolerate 2.4 mg, the label allows extended use of 1.7 mg as a chronic maintenance dose, though weight loss at 1.7 mg averages roughly 1 to 2 percentage points less than at 2.4 mg.
Ozempic (subcutaneous, type 2 diabetes)
Ozempic starts at 0.25 mg weekly for 4 weeks (a starter dose, not therapeutic), then moves to 0.5 mg [12]. Patients can stay there or step up to 1 mg after at least 4 weeks, and to 2 mg if more A1c reduction is needed [23]. The 2 mg dose was added to the label in March 2022 after the SUSTAIN FORTE trial showed an additional 0.23% A1c drop over 1 mg.
Rybelsus (oral, type 2 diabetes)
Rybelsus is dosed daily at 3 mg for 30 days as a tolerance-builder, then 7 mg for at least 30 days, with an option to escalate to 14 mg if A1c targets are not met [22]. The tablet must be swallowed whole on an empty stomach in the morning with no more than 4 ounces of plain water, and nothing else by mouth for at least 30 minutes [22]. Coffee, juice, food, and other oral medications all reduce semaglutide absorption to clinically meaningless levels.
Oral Wegovy (tablet, weight management)
The 25 mg oral Wegovy tablet uses a five-step titration spanning 16 weeks: 3 mg, 7 mg, 14 mg, 17 mg, then 25 mg, each held for 4 weeks [15]. The same fasting administration rules apply as Rybelsus: empty stomach, up to 4 ounces of water, 30-minute wait before food or other medications.
Steady state takes about 5 weeks of weekly dosing because of the long half-life. That cuts both ways. Side effects from a dose increase build slowly and can keep building for a month. If nausea spikes at 1.0 mg, dropping back to 0.5 mg will not bring relief for several weeks, so most clinicians slow the titration rather than reverse it. See the titration schedule guide for clinical pacing strategies.
STEP trials: weight loss in obesity
The Semaglutide Treatment Effect in People with Obesity (STEP) program tested the 2.4 mg dose across eight phase 3 trials enrolling roughly 5,000 adults and 200 adolescents.
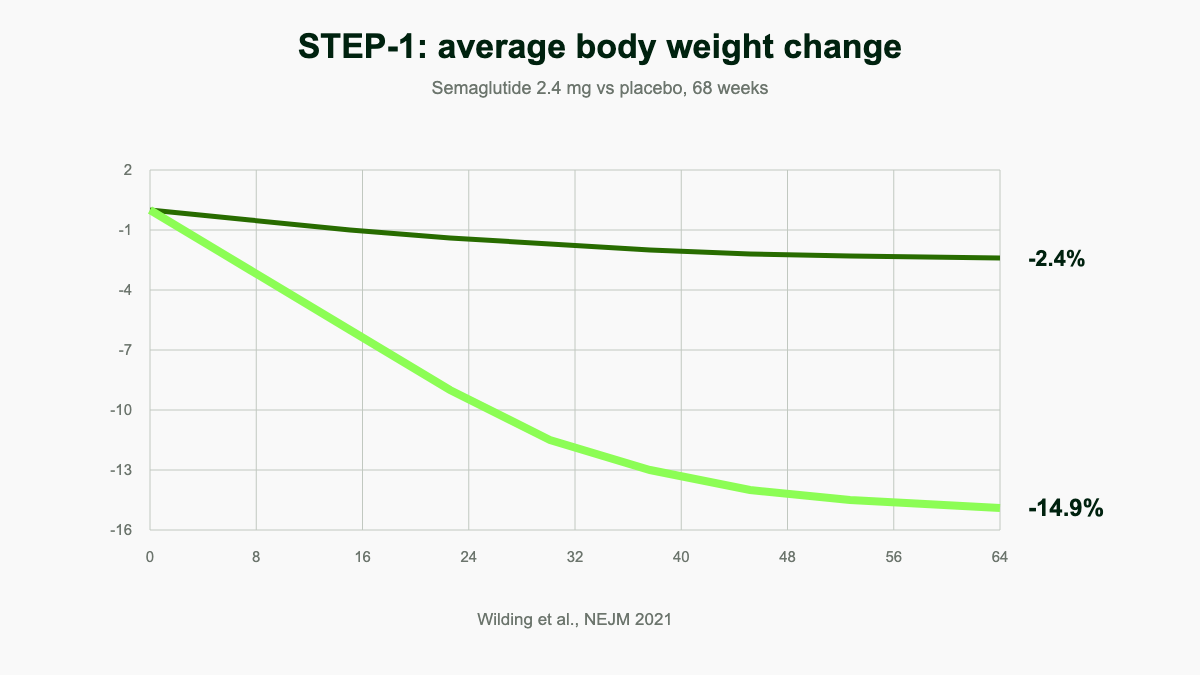
STEP 1 randomized 1,961 adults with obesity but without diabetes to semaglutide 2.4 mg or placebo for 68 weeks. The semaglutide group lost a mean of 14.9% of body weight versus 2.4% for placebo [1]. About 86% of participants on the active drug lost at least 5%, and roughly 32% lost 20% or more [1]. STEP 2 enrolled 1,210 adults with both obesity and type 2 diabetes and showed somewhat smaller losses of 9.6% on the 2.4 mg dose, a pattern seen across most weight-loss trials in diabetic populations [24].
STEP 3 paired semaglutide with intensive behavioral therapy and produced 16.0% mean weight loss at 68 weeks, the highest in the original program [24]. STEP 4 randomized participants who had already lost weight on semaglutide to continue or switch to placebo; those who stayed on semaglutide lost an additional 7.9% from week 20 to week 68, while the placebo-switched group regained 6.9% [25]. STEP 5 extended dosing to 104 weeks and showed sustained 15.2% weight loss versus 2.6% for placebo, confirming that the plateau holds for at least two years [26].
STEP 6 ran in East Asian populations and found 13.2% mean loss versus 2.1% for placebo. STEP 8 directly compared semaglutide 2.4 mg with liraglutide 3.0 mg and showed semaglutide produced 15.8% loss versus 6.4% for liraglutide. STEP TEENS, published in NEJM in November 2022, randomized 201 adolescents aged 12 to 17 with obesity to semaglutide 2.4 mg or placebo for 68 weeks; the semaglutide group lost 16.1% of BMI versus a 0.6% gain for placebo, and 53% achieved at least 15% BMI reduction [27]. Nearly 45% of treated adolescents fell below the obesity BMI cutoff entirely [27].
STEP UP, published in 2025, tested an experimental 7.2 mg weekly dose in adults with obesity and showed 20.7% mean weight loss at 72 weeks versus 17.5% on 2.4 mg, suggesting a dose-response curve that has not yet plateaued.
SUSTAIN trials: glycemic control in type 2 diabetes
The SUSTAIN program comprised six initial phase 3 trials plus four phase 3b extensions, enrolling roughly 10,000 adults with type 2 diabetes [28].
SUSTAIN 1 through 5 tested semaglutide as monotherapy, add-on to metformin, add-on to basal insulin, and against sitagliptin and exenatide. A1c reductions ranged from 1.5% to 1.8% on the 1 mg dose across these trials, with weight loss of 4 to 6 kg [28]. SUSTAIN 6 was the cardiovascular outcomes trial in 3,297 patients with type 2 diabetes at high cardiovascular risk and reported A1c reductions of 1.1% with the 0.5 mg dose and 1.4% with 1 mg, with weight loss of 3.6 kg and 4.9 kg respectively [2].
SUSTAIN 7 compared semaglutide 1 mg with dulaglutide 1.5 mg and showed superior A1c reduction (1.8% versus 1.4%) and superior weight loss (6.5 kg versus 3.0 kg). SUSTAIN 10 compared semaglutide 1 mg with liraglutide 1.2 mg and showed A1c reductions of 1.7% versus 1.0% [29]. SUSTAIN FORTE later showed the 2 mg dose produces an additional 0.23% A1c drop over 1 mg, supporting the March 2022 label addition.
SUSTAIN 6 also tracked cardiovascular outcomes and found a 26% relative reduction in nonfatal stroke, MI, or cardiovascular death (HR 0.74, 95% CI 0.58-0.95) [2]. That trial provided the basis for the January 2020 cardiovascular indication on Ozempic.
SELECT trial: cardiovascular outcomes in obesity
SELECT (Semaglutide Effects on Cardiovascular Outcomes in People With Overweight or Obesity), published in NEJM in November 2023, enrolled 17,604 adults aged 45 or older with overweight or obesity (BMI 27+) and pre-existing cardiovascular disease but no diabetes [3]. The trial ran from October 2018 to June 2023 across 41 countries with median follow-up of 40 months [3].
The primary endpoint, a composite of cardiovascular death, nonfatal MI, or nonfatal stroke, occurred in 6.5% of the semaglutide group and 8.0% of placebo recipients, a 20% relative risk reduction (HR 0.80, 95% CI 0.72-0.90, P<0.001) [3]. All-cause mortality was 19% lower (HR 0.81, 95% CI 0.71-0.93), and heart failure events were 18% lower (HR 0.82, 95% CI 0.71-0.96) [3]. Mean weight loss was 9.4%, smaller than in dedicated obesity trials but still clinically meaningful [3]. SELECT supplied the evidence for the March 2024 cardiovascular indication on Wegovy and made semaglutide the first weight-loss drug ever shown to reduce cardiovascular events.
FLOW trial: chronic kidney disease
FLOW randomized 3,533 adults with type 2 diabetes and chronic kidney disease (eGFR 25-75 ml/min/1.73 m², albuminuria) to semaglutide 1 mg weekly or placebo with median 3.4-year follow-up [30]. The trial was stopped early at a prespecified interim analysis because of clear efficacy.
The primary composite of major kidney disease events (kidney failure, sustained 50% eGFR reduction, or kidney or cardiovascular death) was 24% lower with semaglutide (HR 0.76, 95% CI 0.66-0.88, P=0.0003) [30]. The annual eGFR slope was 1.16 ml/min/1.73 m² less steep, cardiovascular death was 29% lower, and serious adverse events were less common with semaglutide than placebo [30]. FLOW underpinned the January 2025 Ozempic indication for slowing kidney disease progression in type 2 diabetes with CKD.
SOUL trial: cardiovascular outcomes with oral semaglutide
SOUL, published in NEJM in March 2025, enrolled 9,650 adults aged 50 or older with type 2 diabetes (A1c 6.5-10.0%) plus established atherosclerotic cardiovascular disease, chronic kidney disease, or both [31]. Participants received oral semaglutide titrated to 14 mg daily or placebo on top of standard care.
The primary MACE composite occurred in 12.0% of the oral semaglutide group versus 13.8% on placebo, a 14% relative risk reduction (HR 0.86, 95% CI 0.77-0.96, P=0.006) [31]. Serious adverse events did not increase. SOUL supported the October 2025 Rybelsus cardiovascular label expansion and confirmed that the cardioprotection seen with injectable semaglutide extends to the oral formulation.
STEP-HFpEF: heart failure with preserved ejection fraction
STEP-HFpEF and STEP-HFpEF DM together enrolled roughly 1,150 adults with obesity-related heart failure with preserved ejection fraction. Semaglutide 2.4 mg improved Kansas City Cardiomyopathy Questionnaire scores by approximately 7.3 points more than placebo, a clinically large change in heart failure symptoms and physical function [32]. NT-proBNP, a marker of cardiac wall stress, dropped roughly 20% more in the semaglutide arm, and 6-minute walk distance improved by about 20 meters [32].
STRIDE: peripheral artery disease
STRIDE, presented at ACC 2025, randomized 792 adults with type 2 diabetes and symptomatic peripheral artery disease to semaglutide 1 mg weekly or placebo for 52 weeks [33]. The trial was conducted across 112 outpatient clinics in North America, Asia, and Europe from October 2020 to July 2024 [33].
Maximum walking distance improved 13% more with semaglutide than placebo, and disease progression risk fell by 54% [33]. Patients on semaglutide walked a median 26 meters farther on a treadmill at one year [33]. Novo Nordisk filed a label-expansion application with the FDA based on STRIDE.
ESSENCE: noncirrhotic MASH
ESSENCE enrolled 1,197 adults with biopsy-confirmed metabolic dysfunction-associated steatohepatitis and stage 2 or 3 liver fibrosis, randomized 2:1 to weekly semaglutide 2.4 mg or placebo for 240 weeks [34]. At the prespecified 72-week interim, 62.9% of semaglutide recipients achieved resolution of steatohepatitis without worsening of fibrosis versus 34.3% on placebo [34]. Fibrosis improvement without worsening of steatohepatitis occurred in 36.8% versus 22.4% [34]. The data supported the August 2025 FDA approval of Wegovy 2.4 mg for noncirrhotic MASH with moderate to advanced fibrosis.
OASIS and PIONEER: oral programs
The PIONEER program tested oral semaglutide for type 2 diabetes across ten phase 3 trials. PIONEER 1 placebo-adjusted A1c reductions at 26 weeks were 0.6% (3 mg), 0.9% (7 mg), and 1.1% (14 mg), with the 14 mg dose achieving target A1c <7.0% in 55-77% of participants depending on subgroup [35]. PIONEER trials also showed similar A1c reductions to liraglutide and superior reductions to sitagliptin and empagliflozin [35].
The OASIS program tested higher oral doses (25 mg and 50 mg) for weight management. OASIS 4, a 64-week trial in 307 adults with obesity or overweight plus a comorbidity, showed 16.6% mean weight loss with full adherence on the 25 mg daily dose versus 2.7% on placebo [36]. One-third of adherent participants lost at least 20% of body weight [36]. Nausea (46.6% versus 18.6%) and vomiting (30.9% versus 5.9%) were the most common adverse events [36]. OASIS 4 provided the basis for the December 2025 FDA approval of oral Wegovy 25 mg.
Side effects of semaglutide
Most semaglutide side effects are gastrointestinal and most appear during titration. From the Wegovy label: nausea affects roughly 44% of users, diarrhea 30%, vomiting 24%, constipation 24%, abdominal pain 20%, headache 14%, fatigue 11%, dyspepsia 9%, dizziness 8%, and abdominal distension 7% [14]. They tend to peak in the days after a dose increase and fade as the body adapts. Smaller, more frequent meals, avoiding fatty or rich food, and staying hydrated are the standard countermeasures. Roughly 7% of Wegovy users discontinue treatment because of gastrointestinal side effects, mostly within the first three months [14].
Less common but serious adverse events include acute pancreatitis, gallbladder disease (cholelithiasis and cholecystitis), acute kidney injury (usually triggered by dehydration from severe vomiting), and hypoglycemia when semaglutide is combined with insulin or sulfonylureas [12][14]. The FDA added a warning about suicidal thoughts in 2024 after post-marketing reports, though large database studies have not shown an elevated signal. The European Medicines Agency added non-arteritic anterior ischemic optic neuropathy (NAION), a rare cause of sudden vision loss, to the label in 2025 after a Danish-Norwegian cohort found a hazard ratio of 2.81 (incidence rate difference 1.41 per 10,000 person-years) [37]. Absolute risk remains very low, but new monocular vision changes warrant urgent ophthalmology review.
The boxed warning concerns thyroid C-cell tumors. Rats given semaglutide developed medullary thyroid carcinoma at higher rates [12]. Human relevance is unknown, since rodent C-cells express GLP-1 receptors much more densely than human C-cells, but the warning remains and the drug is contraindicated in patients with personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 [12][14].
Who should not take semaglutide
Absolute contraindications are a personal or family history of medullary thyroid carcinoma, multiple endocrine neoplasia type 2 (MEN 2), or a prior serious hypersensitivity reaction to semaglutide [12][14]. Pregnancy is also a contraindication for Wegovy, and patients planning pregnancy should stop the drug at least two months before conception because of the long washout time [14].
Caution is warranted in patients with a history of pancreatitis, severe gastroparesis, active gallbladder disease, or diabetic retinopathy (rapid glucose lowering can transiently worsen retinopathy, an effect seen in SUSTAIN 6) [2]. Semaglutide has not been studied in patients with type 1 diabetes and is not a substitute for insulin.
Special populations
Renal impairment requires no dose adjustment for any of the four products, even in end-stage kidney disease, because elimination is primarily proteolytic [21]. Hepatic impairment likewise requires no adjustment; semaglutide pharmacokinetics did not change meaningfully across mild, moderate, or severe hepatic impairment in dedicated studies [21]. Patients on dialysis can take Ozempic but should be monitored for volume depletion if vomiting or diarrhea develops.
Elderly patients (65+) tolerate semaglutide similarly to younger adults, though sarcopenia risk during weight loss warrants attention. Adolescents 12 and older may receive Wegovy for obesity based on STEP TEENS [27]. Pregnancy and lactation are contraindicated for all four products; semaglutide crosses the placenta in animal studies and produced fetal harm at clinical exposures.
Drug interactions
Because semaglutide slows gastric emptying, oral medications with narrow absorption windows can be affected. The clinically relevant interactions are limited but worth understanding [38][39]:
- Insulin and sulfonylureas: hypoglycemia risk rises sharply; consider 20-30% basal insulin dose reduction at semaglutide initiation
- Warfarin: Tmax is delayed but AUC remains within the therapeutic window; closer INR monitoring is reasonable after dose changes
- Levothyroxine with Rybelsus: oral semaglutide raises levothyroxine exposure by roughly 33%; check TSH 4-6 weeks after initiation [40]
- Oral contraceptives: no clinically relevant exposure change; severe vomiting could still threaten contraceptive absorption
- Other oral medications taken with Rybelsus: any tablet or capsule taken within 30 minutes of Rybelsus has reduced absorption
Alcohol does not directly interact but can worsen nausea and hypoglycemia risk.
Storage, missed dose, and switching
Unopened pens are stored in the refrigerator at 36 to 46 degrees Fahrenheit (2-8 C) [12][14]. Once in use, Ozempic pens last 56 days at room temperature up to 86 F (15-30 C), and Wegovy pens last 28 days [14][41]. Pens are single-patient devices and should never be shared, even with a new needle, because of bloodborne pathogen risk. Ozempic ships with NovoFine Plus 32-gauge, 4 mm needles, which are among the shortest and thinnest available [41].
Injection sites rotate among the abdomen, thigh, and upper arm. The day of the week can be changed as long as at least 48 hours have passed since the last dose. A missed dose can be taken within 5 days of the scheduled day; if more than 5 days have passed, skip it and resume on the regular schedule [12].
Switching between brands requires care. From Ozempic 1 mg to Wegovy: most clinicians restart titration at 1.0 mg Wegovy and step up to 2.4 mg over 12 weeks. From semaglutide to tirzepatide or vice versa: a 1-week washout is generally enough given the matching half-lives. From Rybelsus to Ozempic: stop the tablet the day the first injection is given. See the switching guide for protocol-level detail.
Cost and access
The list price for Ozempic is approximately $968 per month and Wegovy is approximately $1,349 per month [18]. Insurance coverage varies dramatically: most commercial plans cover Ozempic for type 2 diabetes, while Wegovy obesity coverage remains spotty. Medicare cannot cover anti-obesity medications by statute, though Wegovy gained Medicare Part D coverage for the cardiovascular indication in March 2024 after the SELECT label expansion.
Novo Nordisk runs a savings card for commercially insured patients that can cap copay at $25 per month for Wegovy with a $200 monthly maximum savings [42]. The NovoCare Pharmacy direct-to-patient channel sells Wegovy at $499 per month for the 2.4 mg maintenance dose to cash-paying patients without insurance, with starter-dose pricing at $199 through June 2026 [19][42]. Government beneficiaries (Medicare, Medicaid, TRICARE) are excluded from the commercial savings card. The Patient Assistance Program covers free medication for low-income patients but only for diabetes indications, not obesity.
Compounded semaglutide
The FDA listed semaglutide on the drug shortage list from 2022 through February 2025, which permitted compounding pharmacies to make non-FDA-approved versions under Sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act [43]. The shortage resolved on February 21, 2025, ending the broad enforcement window. Section 503A compounders had until April 22, 2025, and Section 503B outsourcing facilities until May 22, 2025, to wind down semaglutide production from bulk substances [43].
Since June 17, 2025, compounded semaglutide can only be dispensed under a patient-specific medical-necessity exemption documenting why the FDA-approved version cannot be used (allergy to an inactive ingredient, dose not commercially available, etc.) [43]. The FDA has also proposed to formally exclude semaglutide from the 503B bulks list, citing no clinical need for outsourcing-facility compounding [43]. See compounded vs brand for a detailed comparison.
Future indications under study
Semaglutide is being tested in dozens of phase 2 and phase 3 trials beyond its current label. The EVOKE and EVOKE+ trials enrolled 3,808 adults with mild cognitive impairment or mild Alzheimer's dementia and reported topline results in November 2025: oral semaglutide 14 mg did not slow clinical progression on the CDR-SB primary endpoint at week 104, though CSF p-tau181 dropped 10% compared with placebo, suggesting some biomarker effect without clinical benefit [44].
Other ongoing programs include semaglutide for alcohol and opioid use disorders (multiple academic trials underway), polycystic ovary syndrome, idiopathic intracranial hypertension, knee osteoarthritis (the STEP 9 trial showed pain and function improvements proportional to weight loss), and cardiovascular protection in primary prevention. A monthly subcutaneous formulation is in early-phase development.
Practical takeaways
Semaglutide is the most studied and most widely used GLP-1 receptor agonist in the world, with more than 25 million patient-years of cumulative exposure and outcomes data spanning glycemic control, weight management, cardiovascular protection, kidney protection, heart failure, peripheral artery disease, and liver disease. The three brands serve distinct goals at distinct doses, and the choice between Ozempic, Wegovy, and Rybelsus comes down to indication, route preference, and insurance coverage rather than active drug.
Most patients should expect a 16-week titration to maintenance dose, manageable but persistent gastrointestinal side effects, and clinically meaningful weight loss or A1c reduction by month four. Those starting therapy should plan ahead for refrigeration, set up a savings card or direct-pay channel before the first refill, and schedule a 12-week follow-up to review tolerability and dose adequacy.
References
- Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1). New England Journal of Medicine, 2021. nejm.org
- Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (SUSTAIN-6). New England Journal of Medicine, 2016. nejm.org
- Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity Without Diabetes (SELECT). New England Journal of Medicine, 2023. nejm.org
- Drugs.com. Semaglutide: Uses, Dosage, Side Effects, Brands. Drugs.com, 2026. drugs.com
- U.S. National Library of Medicine. Semaglutide Injection: MedlinePlus Drug Information. MedlinePlus, 2026. medlineplus.gov
- Mayo Clinic. Semaglutide (Subcutaneous Route): Description and Brand Names. Mayo Clinic, 2026. mayoclinic.org
- Novo Nordisk. Wegovy (semaglutide) Prescribing Information and Patient Site. Novo Nordisk, 2026. wegovy.com
- Novo Nordisk. Ozempic (semaglutide) Prescribing Information and Patient Site. Novo Nordisk, 2026. ozempic.com
- WebMD. Ozempic (Semaglutide): Uses, Side Effects, Interactions. WebMD, 2026. webmd.com
- Wikipedia contributors. Semaglutide. Wikipedia, 2026. wikipedia.org
- Hall S, Isaacs D, Clements JN. Pharmacokinetics and Clinical Implications of Semaglutide: A New Glucagon-Like Peptide (GLP)-1 Receptor Agonist. Clinical Pharmacokinetics, 2018. ncbi.nlm.nih.gov
- U.S. Food & Drug Administration. Ozempic (semaglutide) Injection Prescribing Information, NDA 209637. accessdata.fda.gov, 2025. accessdata.fda.gov
- U.S. Food & Drug Administration. Rybelsus (semaglutide) Tablets Prescribing Information, NDA 213051. accessdata.fda.gov, 2025. accessdata.fda.gov
- U.S. Food & Drug Administration. Wegovy (semaglutide) Injection Prescribing Information, NDA 215256. accessdata.fda.gov, 2025. accessdata.fda.gov
- Pharmaceutical Executive. FDA Approves Novo Nordisk's Wegovy Pill for Weight Management. PharmExec, December 2025. pharmexec.com
- U.S. Food & Drug Administration. FDA Approves Treatment for Serious Liver Disease Known as MASH. FDA News, August 15, 2025. fda.gov
- Prime Therapeutics. GLP-1 Pipeline Update: 2025. Prime Therapeutics, 2025. primetherapeutics.com
- Drugs.com. Semaglutide Prices and Coupons. Drugs.com, 2026. drugs.com
- NovoCare. Wegovy Self-Pay through NovoCare Pharmacy. Novo Nordisk, 2026. novocare.com
- American Journal of Managed Care. FDA Approves Semaglutide to Prevent Heart Events in Patients With CVD and Excess Weight. AJMC, 2024. ajmc.com
- Jensen L, Helleberg H, Roffel A, et al. Pharmacokinetics and Tolerability of Semaglutide in People with Hepatic Impairment. Diabetes, Obesity and Metabolism, 2018. ncbi.nlm.nih.gov
- U.S. Food & Drug Administration. Rybelsus Tablets, Highlights of Prescribing Information. accessdata.fda.gov, 2024. accessdata.fda.gov
- Frias JP, Auerbach P, Bajaj HS, et al. Efficacy and Safety of Once-Weekly Semaglutide 2.0 mg vs 1.0 mg in Type 2 Diabetes (SUSTAIN FORTE). Lancet Diabetes & Endocrinology, 2021.
- Ghusn W, De la Rosa A, Sacoto D, et al. Weight Loss Outcomes Associated With Semaglutide Treatment for Patients With Overweight or Obesity. JAMA Network Open, 2022. jamanetwork.com
- Rubino D, Abrahamsson N, Davies M, et al. Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance: The STEP 4 Randomized Clinical Trial. JAMA, 2021. jamanetwork.com
- Garvey WT, Batterham RL, Bhatta M, et al. Two-Year Effects of Semaglutide in Adults with Overweight or Obesity: The STEP 5 Trial. Nature Medicine, 2022. nature.com
- Weghuber D, Barrett T, Barrientos-Perez M, et al. Once-Weekly Semaglutide in Adolescents with Obesity (STEP TEENS). New England Journal of Medicine, 2022. nejm.org
- Aroda VR, Ahmann A, Cariou B, et al. Comparative Efficacy, Safety, and Cardiovascular Outcomes with Once-Weekly Subcutaneous Semaglutide in the Treatment of Type 2 Diabetes: Insights from the SUSTAIN 1-7 Trials. Diabetes & Metabolism, 2019. pubmed.ncbi.nlm.nih.gov
- Capehorn MS, Catarig AM, Furberg JK, et al. Efficacy and Safety of Once-Weekly Semaglutide 1.0 mg vs Once-Daily Liraglutide 1.2 mg (SUSTAIN 10). Diabetes & Metabolism, 2020. pubmed.ncbi.nlm.nih.gov
- Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes (FLOW). New England Journal of Medicine, 2024. nejm.org
- McGuire DK, Marx N, Mulvagh SL, et al. Oral Semaglutide and Cardiovascular Outcomes in High-Risk Type 2 Diabetes (SOUL). New England Journal of Medicine, 2025. nejm.org
- Kosiborod MN, Abildstrom SZ, Borlaug BA, et al. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity (STEP-HFpEF). New England Journal of Medicine, 2023. nejm.org
- Bonaca MP, Catarig AM, Houlind K, et al. Semaglutide and Walking Capacity in People with Symptomatic Peripheral Artery Disease and Type 2 Diabetes (STRIDE). The Lancet, 2025. thelancet.com
- Newsome PN, Sanyal AJ, Engebretsen KA, et al. Phase 3 Trial of Semaglutide in Metabolic Dysfunction-Associated Steatohepatitis (ESSENCE). New England Journal of Medicine, 2025. pubmed.ncbi.nlm.nih.gov
- Aroda VR, Blonde L, Pratley RE. Efficacy of Oral Semaglutide: Overview of the PIONEER Clinical Trial Program. American Journal of Managed Care, 2021. ajmc.com
- Knop FK, Aroda VR, do Vale RD, et al. Oral Semaglutide at a Dose of 25 mg in Adults with Overweight or Obesity (OASIS 4). New England Journal of Medicine, 2025. nejm.org
- Hathaway JT, Shah MP, Hathaway DB, et al. Risk of Nonarteritic Anterior Ischemic Optic Neuropathy in Patients Prescribed Semaglutide. JAMA Ophthalmology, 2024. jamanetwork.com
- Endocrinology Advisor. GLP-1 Interactions: Risks, Effects, and Recommendations. Endocrinology Advisor, 2025. endocrinologyadvisor.com
- GoodRx. 7 Rybelsus Drug Interactions to Know Before Taking It. GoodRx, 2026. goodrx.com
- Hauge C, Breitschaft A, Hartoft-Nielsen ML, et al. Effect of Oral Semaglutide on the Pharmacokinetics of Thyroxine after Dosing of Levothyroxine. Expert Opinion on Drug Metabolism & Toxicology, 2021. tandfonline.com
- Novo Nordisk. How to Use Ozempic Pen: Dosing and Storage. Ozempic.com, 2026. ozempic.com
- NovoCare. Savings Offer Program for Wegovy. Novo Nordisk, 2026. novocare.com
- U.S. Food & Drug Administration. FDA Clarifies Policies for Compounders as National GLP-1 Supply Begins to Stabilize. FDA Drug Alerts, 2025. fda.gov
- Cummings JL, Atri A, Feldman HH, et al. Efficacy and Safety of Oral Semaglutide 14 mg in Early-Stage Symptomatic Alzheimer's Disease (evoke and evoke+). The Lancet, 2026. thelancet.com