Tirzepatide (Mounjaro, Zepbound)
Summary: Tirzepatide is a once-weekly injectable that activates both the GIP and GLP-1 receptors. Lilly sells it as Mounjaro for type 2 diabetes and as Zepbound for chronic weight management and obstructive sleep apnea, with doses titrated from 2.5 mg up to 15 mg.
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.
What tirzepatide is
Tirzepatide is a synthetic 39-amino-acid peptide engineered to bind two incretin receptors at the same time: the receptor for glucose-dependent insulinotropic polypeptide (GIP) and the receptor for glucagon-like peptide-1 (GLP-1). The peptide backbone is acylated with a C20 fatty diacid linked through a glutamic acid spacer and two short polyethylene glycol units to a lysine at position 20, an arrangement borrowed from the same albumin-binding playbook that gave semaglutide its long half-life [1][9]. Eli Lilly developed the molecule under the code name LY3298176 and markets it commercially under two brand names that share an identical active ingredient.
Mounjaro received FDA approval on 13 May 2022 for adults with type 2 diabetes as an adjunct to diet and exercise [1]. Zepbound followed on 8 November 2023 for chronic weight management in adults with obesity (body mass index 30 kg/m2 or higher) or overweight (BMI 27 kg/m2 or higher) plus at least one weight-related comorbidity such as hypertension, dyslipidemia, type 2 diabetes, obstructive sleep apnea, or cardiovascular disease [2]. On 20 December 2024 the FDA expanded the Zepbound label to cover moderate-to-severe obstructive sleep apnea in adults with obesity, making tirzepatide the first drug ever approved for that indication [3].
Both Mounjaro and Zepbound contain the same six tirzepatide dose strengths in the same single-dose autoinjector pens, plus single-dose vials for Zepbound. The split exists for regulatory, payer, and supply chain reasons rather than chemical ones. Plans that cover diabetes drugs but exclude obesity drugs will pay for Mounjaro but reject Zepbound, even though the syringes deliver the same molecule at the same milligrams.
How the dual GIP/GLP-1 agonist works
Most marketed incretin drugs, including semaglutide, dulaglutide, and liraglutide, target only the GLP-1 receptor. Tirzepatide adds full agonism at the GIP receptor on top of strong GLP-1 receptor activity, a combination called twincretin pharmacology [9]. The two arms produce overlapping but distinct effects.
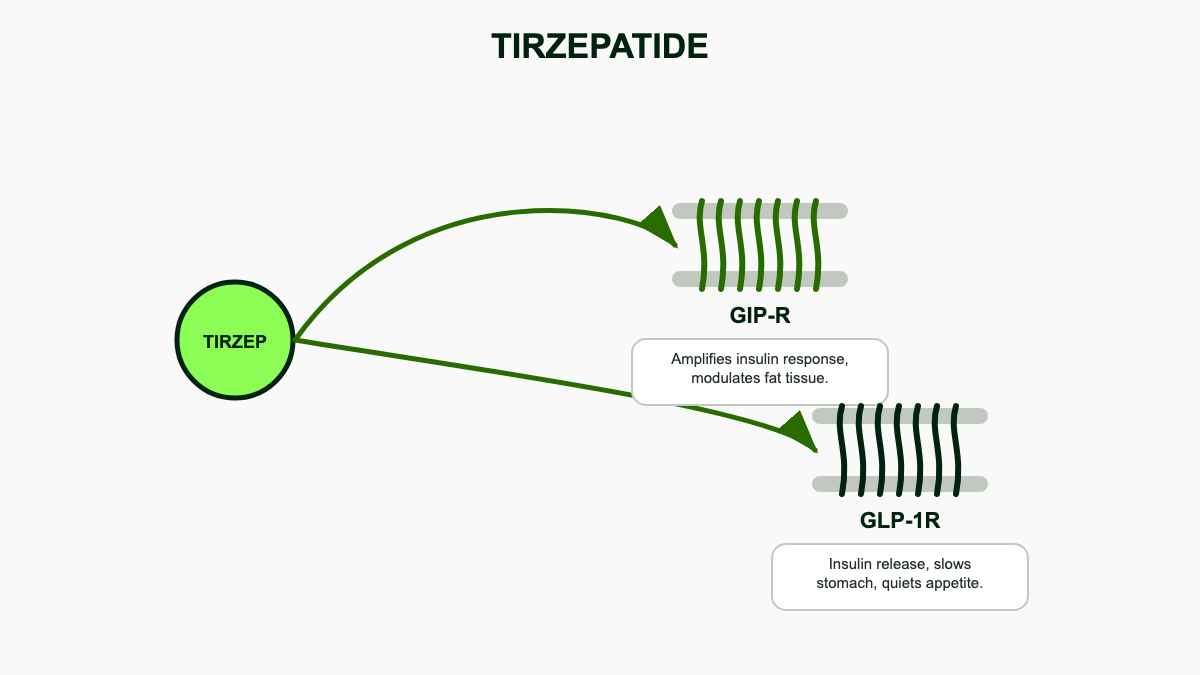
The GLP-1 arm slows gastric emptying, blunts post-meal glucose spikes, dampens appetite-driving circuits in the arcuate nucleus and brainstem, and stimulates insulin release from pancreatic beta cells in a glucose-dependent fashion that limits hypoglycemia risk. It also suppresses glucagon when blood sugar is high. The GIP arm potentiates insulin secretion synergistically with GLP-1 at the beta cell, improves insulin sensitivity in adipose tissue, may increase lipid storage capacity in healthy fat depots while suppressing ectopic fat, and seems to blunt the nausea signal generated by central GLP-1 activation [9]. Animal data also suggest GIP receptor activity in hypothalamic neurons reinforces appetite suppression through a non-overlapping circuit.
Net result: at matched plasma concentrations, tirzepatide drops weight harder than pure GLP-1 agonism while delivering proportionally less gastrointestinal toxicity per kilogram lost. SURMOUNT-5 made that translation concrete by reporting roughly half the rate of GI-driven discontinuations on tirzepatide compared with semaglutide despite larger weight loss [10].
Pharmacokinetics and metabolism
Subcutaneous bioavailability sits around 80 percent. Peak plasma concentration occurs 8 to 72 hours after injection, and the elimination half-life of about 5 days supports once-weekly dosing while keeping trough concentrations within range of the steady-state peak [9]. Steady state is reached after roughly 4 weeks at any given dose, which is why the label spaces dose escalations 4 weeks apart. The volume of distribution at steady state is approximately 10.3 liters, consistent with mostly extracellular distribution, and the molecule is 99 percent protein-bound to albumin.
Tirzepatide is metabolized by proteolytic cleavage of the peptide backbone, beta-oxidation of the C20 fatty diacid side chain, and amide hydrolysis. Metabolites are excreted in urine and feces. There is no involvement of the cytochrome P450 system, so drug-drug interactions on the metabolism side are minimal. Clinical interactions instead come from the pharmacodynamic delay in gastric emptying, which can change absorption of co-administered oral medications.
Drug interactions clinicians actually need to track
The label flags two interaction categories. First, oral contraceptives that depend on stable absorption can lose effectiveness for 4 weeks after starting tirzepatide and after each dose escalation. Patients who use combined oral contraceptives should switch to a non-oral method or add a barrier method during those windows [4][6]. Second, when tirzepatide is added to insulin or sulfonylureas, hypoglycemia risk climbs and clinicians typically reduce the insulin or secretagogue dose by 20 percent or more before the first injection.
Other GLP-1 receptor agonists should not be given alongside tirzepatide because the receptor pharmacology overlaps and risk compounds without added benefit. Drugs with narrow therapeutic indices that depend on rapid absorption (warfarin, levothyroxine, antiepileptics, some immunosuppressants) deserve closer monitoring during titration because the slowed gastric emptying can flatten and delay their absorption curve.
Dosing and titration
Tirzepatide ships in six strengths: 2.5, 5, 7.5, 10, 12.5, and 15 mg, each in a 0.5 mL fixed-dose pen or vial [4][6]. The label requires a stepwise climb to limit gastrointestinal side effects.
- Weeks 1 to 4: 2.5 mg once weekly. This is a starter dose, not a treatment dose.
- Weeks 5 to 8: 5 mg once weekly. First true maintenance option.
- Weeks 9 to 12: 7.5 mg once weekly if further glucose or weight reduction is needed.
- Weeks 13 to 16: 10 mg once weekly.
- Weeks 17 to 20: 12.5 mg once weekly.
- Weeks 21 onward: 15 mg once weekly, the maximum recommended dose.
Clinicians do not need to push every patient to 15 mg. Many people reach their A1c or weight target at 5, 7.5, or 10 mg and stay there indefinitely. The general principle is to advance only after 4 weeks at the prior dose, only if tolerability is acceptable, and only if the clinical goal is unmet.
The injection goes subcutaneously into the abdomen, thigh, or upper arm. The injection site should be rotated each week to limit lipohypertrophy and local irritation. Patients can change which day of the week they dose as long as at least 3 days pass between shots, and a missed dose can be administered within 4 days of the scheduled day; beyond that, skip the dose and resume the regular schedule [4][6]. There is no need to double up.
If treatment is interrupted for more than 6 weeks, most clinicians restart at 2.5 mg and re-titrate to limit recurrence of severe nausea, since gastrointestinal tolerance fades during a long break.
Storage
Refrigerate at 36 to 46 degrees Fahrenheit (2 to 8 degrees Celsius). Pens or vials left at room temperature (below 86 degrees Fahrenheit, or 30 degrees Celsius) are good for up to 21 days, after which they must be discarded [4][6]. Do not freeze, and do not return a pen to the fridge after it has warmed up. Protect from direct sunlight. The vial presentation, used primarily through the LillyDirect self-pay pathway for Zepbound, is a single-dose vial that requires the user to draw up the full 0.5 mL with a separate syringe.
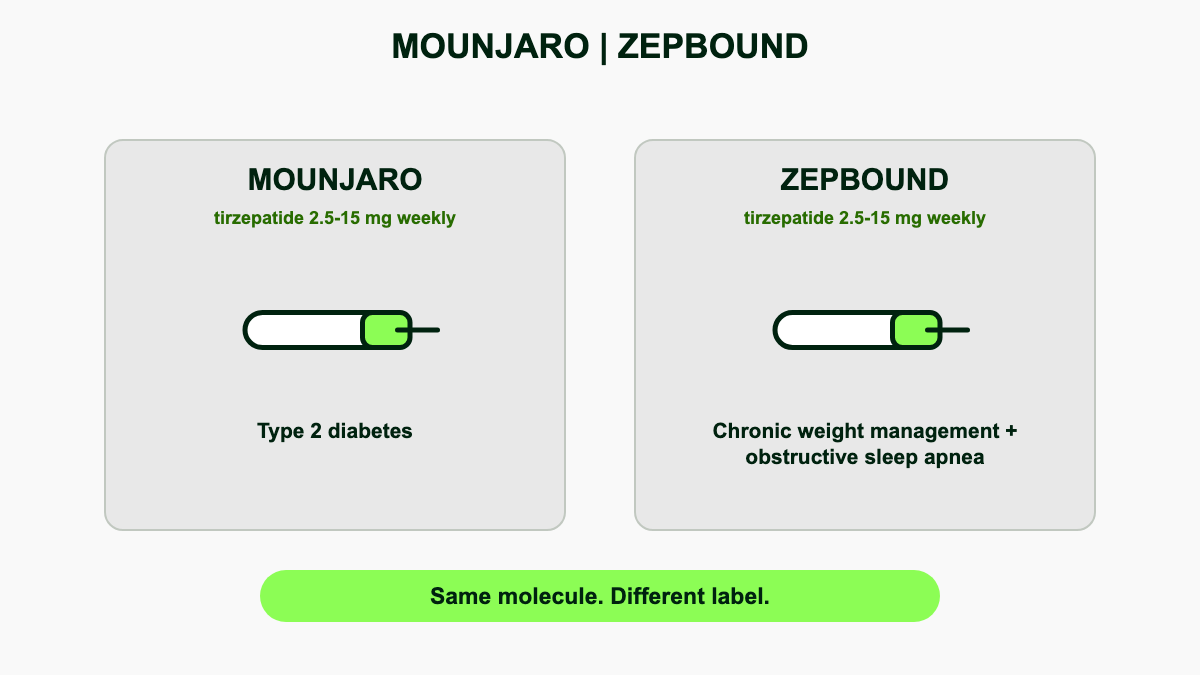
Mounjaro versus Zepbound: same molecule, different paperwork
The cleanest mental model is that tirzepatide is one drug with two FDA labels. Mounjaro is the diabetes label, Zepbound is the obesity and OSA label. Practical differences sit in three areas.
- Insurance coverage. Most US commercial plans cover Mounjaro for type 2 diabetes with prior authorization. Zepbound coverage is patchy because many plans still exclude obesity drugs, even after the OSA expansion. Medicare Part D cannot cover weight-loss indications under current statute, so Medicare beneficiaries who need tirzepatide for obesity alone often pay cash, while Mounjaro is covered for diabetes [13].
- Pricing. List prices for both pen presentations sit near 1,080 to 1,090 dollars per 28-day supply [13]. Lilly's Mounjaro Savings Card can drop commercial-insurance copays as low as 25 dollars per fill for eligible patients. Zepbound's manufacturer support runs through the LillyDirect Self Pay program, which sells single-dose vials at 349 dollars (2.5 mg starter), 499 dollars (5 mg), 599 dollars (7.5 mg), and 699 dollars (10 mg and above) per month directly to consumers without requiring insurance [13].
- Eligibility paperwork. Mounjaro prescriptions require a documented type 2 diabetes diagnosis. Zepbound requires either a BMI 30 or higher or a BMI 27 or higher with a weight-related comorbidity, or a documented diagnosis of moderate-to-severe OSA with obesity for the new sleep apnea indication [2][3].
SURMOUNT trials: weight loss in obesity
The SURMOUNT phase 3 program tested tirzepatide for chronic weight management across diverse populations. The headline numbers from these trials drove the Zepbound approval and reset expectations for what pharmacotherapy can achieve in obesity.
SURMOUNT-1
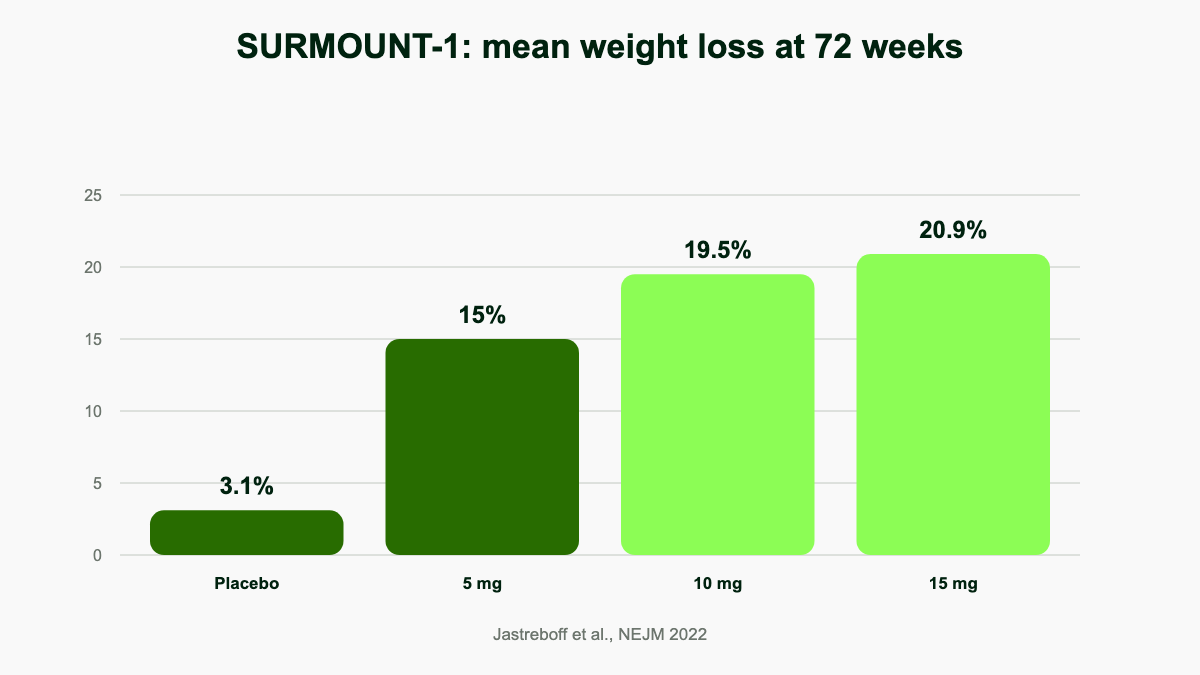
SURMOUNT-1 enrolled 2,539 adults with obesity but without type 2 diabetes (mean baseline BMI 38, mean weight 105 kg) and randomized them to placebo or 5, 10, or 15 mg of tirzepatide for 72 weeks [7]. Mean body-weight change versus baseline:
- Placebo: -3.1 percent
- Tirzepatide 5 mg: -15.0 percent
- Tirzepatide 10 mg: -19.5 percent
- Tirzepatide 15 mg: -20.9 percent
About 50 percent of participants on 10 mg and 57 percent on 15 mg lost at least 20 percent of starting body weight, a threshold that previously belonged almost exclusively to bariatric surgery. The 36 percent of participants in the 15 mg arm who lost 25 percent or more crossed into territory comparable to sleeve gastrectomy outcomes at one year. A pre-diabetes substudy followed for 176 weeks reported a 94 percent reduction in progression to type 2 diabetes among participants who stayed on tirzepatide compared with placebo [9].
SURMOUNT-2
SURMOUNT-2 enrolled 938 adults with obesity and type 2 diabetes. Tirzepatide 10 mg produced 12.8 percent mean weight loss and 15 mg produced 14.7 percent at 72 weeks, versus 3.2 percent on placebo. Diabetes blunts weight-loss responses to incretins by 5 to 7 percentage points relative to non-diabetic populations, but the absolute losses still exceeded any prior obesity drug studied in this group.
SURMOUNT-3
SURMOUNT-3 used a 12-week intensive lifestyle lead-in, then randomized only the 579 participants who had already lost at least 5 percent on lifestyle alone to tirzepatide titrated to maximum tolerated dose or placebo for an additional 72 weeks. The tirzepatide group achieved an additional 18.4 percent weight loss on top of the lifestyle loss, while the placebo group regained 2.5 percent, demonstrating that the drug works as an additive layer rather than a substitute for behavior change.
SURMOUNT-4
SURMOUNT-4 followed 670 adults through a 36-week open-label tirzepatide lead-in (mean weight loss 20.9 percent), then randomized them to continued tirzepatide or placebo for 52 weeks. The continuation group lost an additional 5.5 percent. The withdrawal group regained 14.0 percent, ending up only 9.9 percent below their original baseline. The trial confirmed what most clinicians now treat as fact: stopping tirzepatide reverses most of its benefit within a year, so chronic dosing is the working model.
SURMOUNT-5
SURMOUNT-5, published in the New England Journal of Medicine in May 2025, was the long-awaited head-to-head trial of tirzepatide versus semaglutide for obesity. The open-label phase 3b study randomized 751 adults with obesity but without diabetes to maximum tolerated tirzepatide (10 or 15 mg) or maximum tolerated semaglutide (1.7 or 2.4 mg) for 72 weeks [10]. Mean weight loss was 20.2 percent on tirzepatide versus 13.7 percent on semaglutide, a 6.5 percentage point absolute difference (P less than 0.001). Tirzepatide also won on waist circumference reduction (-18.4 cm versus -13.0 cm) and on the proportion of participants reaching weight-loss thresholds of 10, 15, 20, and 25 percent. Notably, gastrointestinal adverse events leading to discontinuation occurred in 2.7 percent of the tirzepatide group versus 5.6 percent of the semaglutide group, half the rate, undermining the assumption that bigger weight loss must come with more GI cost.
SURMOUNT-OSA
SURMOUNT-OSA enrolled 469 adults with moderate-to-severe obstructive sleep apnea (apnea-hypopnea index 15 events/hour or higher) and obesity, split into two intervention-specific appendices: ISA-1 enrolled people not using positive airway pressure therapy and ISA-2 enrolled people already on PAP [11]. After 52 weeks at maximum tolerated tirzepatide:
- ISA-1 (no PAP): AHI dropped by 25.3 events/hour on tirzepatide versus 5.3 events/hour on placebo, a treatment difference of -20.0 events/hour. Body weight fell 16.1 percent.
- ISA-2 (on PAP): AHI dropped by 29.3 events/hour versus 5.5 events/hour on placebo, a treatment difference of -23.8 events/hour. Body weight fell 17.3 percent.
About 50 percent of the tirzepatide arm achieved disease resolution or mild residual disease (AHI under 5 events/hour, or AHI 5 to 14 with no excessive daytime sleepiness). The trial also reported improvements in patient-reported outcomes including daytime sleepiness, fatigue, and sleep-related quality of life. These results led to the December 2024 FDA approval of Zepbound as the first drug indicated for obstructive sleep apnea [3].
SURPASS trials: type 2 diabetes
The SURPASS phase 3 program tested Mounjaro against placebo, basal insulin, and an active GLP-1 comparator across six trials [12].
- SURPASS-1 (40 weeks, monotherapy vs placebo, n=478): A1c dropped from 7.95 to 5.88 on the 15 mg arm, with weight loss of 11.0 percent. Placebo arm A1c rose slightly.
- SURPASS-2 (40 weeks, vs semaglutide 1 mg, n=1,879): A1c fell from 8.38 to 5.82 on tirzepatide 15 mg versus 6.42 on semaglutide 1 mg [8]. Mean weight loss reached 11.2 kg on 15 mg versus 5.7 kg on semaglutide. The proportion of patients reaching A1c under 7 percent was about 86 percent on tirzepatide 15 mg versus 79 percent on semaglutide.
- SURPASS-3 (52 weeks, vs insulin degludec, n=1,444): A1c fell from 8.18 to 5.81 on 15 mg versus 6.85 on insulin degludec. Tirzepatide produced 13.9 percent weight loss while degludec patients gained roughly 2.6 kg.
- SURPASS-4 (52 weeks, vs insulin glargine in patients with elevated cardiovascular risk, n=2,002): A1c reductions of 2.43 percent on 15 mg versus 1.44 percent on glargine, with weight loss of 11.7 kg versus weight gain of 1.9 kg. A pre-specified cardiovascular meta-analysis showed no excess major adverse cardiac events.
- SURPASS-5 (40 weeks, add-on to insulin glargine vs placebo, n=475): A1c dropped 2.34 percent on 15 mg from a baseline of 8.31 percent, reaching 5.73 percent. Weight loss was 11.6 percent at 15 mg.
- SURPASS-6 (52 weeks, vs prandial insulin lispro added to basal insulin, n=1,428): A1c fell 2.1 percent on tirzepatide versus 1.1 percent on lispro, and tirzepatide patients lost 9 kg while lispro patients gained 4 kg.
Across the program, severe hypoglycemia was rare on tirzepatide unless background insulin or sulfonylurea was present, and most A1c lowering came in the first 24 weeks before plateauing.
SUMMIT: heart failure with preserved ejection fraction
The SUMMIT trial published in the New England Journal of Medicine in November 2024 randomized 731 adults with obesity-related HFpEF (BMI 30 or higher, ejection fraction 50 percent or higher) to tirzepatide titrated to 15 mg or placebo for a median 104 weeks [14]. The primary composite endpoint of cardiovascular death or worsening heart failure occurred in 9.9 percent of the tirzepatide arm versus 15.3 percent of placebo, a hazard ratio of 0.62 (95 percent CI 0.41 to 0.95, P=0.026). Worsening heart failure events alone fell from 14.2 percent to 8.0 percent (HR 0.54). The Kansas City Cardiomyopathy Questionnaire clinical summary score improved 6.9 points more on tirzepatide than on placebo (P less than 0.001). Tirzepatide also produced larger reductions in body weight, waist circumference, NT-proBNP, high-sensitivity CRP, and systolic blood pressure. SUMMIT positions tirzepatide as one of a handful of treatments with mortality-relevant signals in HFpEF and is driving discussions about a potential cardiovascular indication.
SYNERGY-NASH: metabolic dysfunction-associated steatohepatitis
SYNERGY-NASH was a phase 2 trial of 190 adults with biopsy-confirmed MASH (formerly called NASH) and stage F2 or F3 fibrosis, randomized to tirzepatide 5, 10, or 15 mg or placebo for 52 weeks [15]. MASH resolution without worsening of fibrosis at 52 weeks was achieved in 44 percent of the 5 mg group, 56 percent of the 10 mg group, and 62 percent of the 15 mg group, versus 10 percent on placebo. At least one-stage fibrosis improvement without MASH worsening occurred in 51 to 55 percent of tirzepatide arms versus 30 percent on placebo. The phase 3 SYNERGY-NASH-2 trial is enrolling.
Tirzepatide versus semaglutide
The pragmatic question for most patients is whether to use tirzepatide or semaglutide (Ozempic for diabetes, Wegovy for obesity). The data so far favors tirzepatide on raw efficacy.
- Glycemic control: Modestly better A1c reduction at all matched doses in SURPASS-2 [8]. Tirzepatide 15 mg dropped A1c about 0.5 percentage points more than semaglutide 1 mg.
- Weight loss: 20.2 percent on tirzepatide versus 13.7 percent on semaglutide at 72 weeks in the SURMOUNT-5 head-to-head trial, an absolute 6.5 percentage point advantage [10].
- Tolerability: SURMOUNT-5 found discontinuation for GI adverse events was 2.7 percent on tirzepatide versus 5.6 percent on semaglutide, suggesting tirzepatide is at least as well tolerated despite producing more weight loss [10].
- Mechanism: Tirzepatide adds GIP agonism; semaglutide acts at GLP-1 only.
- Cardiovascular outcomes evidence: Semaglutide has the SUSTAIN-6 and SELECT cardiovascular outcomes trials in its label. Tirzepatide does not yet have a dedicated CV outcomes trial reported, though SUMMIT and the SURPASS-CVOT trial currently underway will fill that gap.
Cost, insurance coverage, and supply availability often decide the prescription in practice. Patients on stable semaglutide who are at goal generally do not need to switch.
Side effects
The dominant adverse events are gastrointestinal and dose-related, with frequencies pulled from the pooled SURMOUNT and SURPASS programs [4][5][6].
- Nausea (12 to 33 percent across doses, peaking after each escalation)
- Diarrhea (12 to 23 percent)
- Constipation (6 to 17 percent)
- Vomiting (5 to 13 percent)
- Abdominal pain (5 to 10 percent), dyspepsia, bloating, eructation
- Decreased appetite (5 to 11 percent), generally a desired effect
- Injection-site reactions (3 percent)
- Fatigue (4 to 7 percent), hair shedding (5 percent in obesity trials, usually temporary), hypersensitivity reactions (rare)
Most GI symptoms are mild to moderate and concentrate in the first 4 to 8 weeks of any new dose. Practical mitigation: eat smaller meals, stop before fullness, drop fatty and fried foods, slow dose escalation if symptoms break through, and consider holding at the current dose for an extra 4 weeks before climbing again.
Less common but more serious risks include:
- Acute pancreatitis, signaled by severe persistent abdominal pain that may radiate to the back, with or without vomiting. Stop the drug and evaluate immediately.
- Gallbladder disease, including cholelithiasis and cholecystitis. Rapid weight loss is the underlying mechanism more than direct drug toxicity. Reported in roughly 0.6 to 1.7 percent of treated patients.
- Acute kidney injury, almost always secondary to dehydration from severe vomiting or diarrhea. Patients with chronic kidney disease should be counseled on hydration.
- Hypoglycemia, primarily when tirzepatide is combined with insulin or a sulfonylurea. Rare as monotherapy in people without diabetes.
- Diabetic retinopathy progression in people with pre-existing eye disease, possibly related to rapid glycemic improvement. Baseline retinal exam is reasonable in patients with long-standing or poorly controlled diabetes [5].
- Anaphylaxis and angioedema, rare but reported in post-marketing surveillance.
- Suicidal ideation has been investigated as a class signal for incretin drugs; FDA reviews to date have not confirmed a causal association, but clinicians should ask about mood changes during follow-up.
- Aspiration risk during anesthesia. Slowed gastric emptying may leave food in the stomach beyond standard fasting windows. American Society of Anesthesiologists guidance recommends holding the weekly dose for at least 1 week before elective procedures requiring sedation.
Estrogen-containing oral contraceptives can lose effectiveness for 4 weeks after starting tirzepatide and after each dose increase because slowed gastric emptying alters absorption. The label recommends a non-oral backup or alternative method during that window [4].
Boxed warning and contraindications
Tirzepatide carries a boxed warning for thyroid C-cell tumors based on rodent studies in which similar long-acting GLP-1 agonists produced thyroid C-cell tumors at clinically relevant exposures [4][5]. Whether this risk translates to humans is unknown, with no clear signal in pooled clinical trial data, but caution remains mandatory.
Do not use tirzepatide if any of the following apply:
- Personal or family history of medullary thyroid carcinoma
- Multiple endocrine neoplasia syndrome type 2 (MEN 2)
- Prior serious hypersensitivity to tirzepatide or any component of the formulation
Tirzepatide has not been adequately studied in pregnancy and is not recommended. Animal reproductive studies showed embryofetal harm at clinically relevant exposures. Women planning conception should stop the drug at least 2 months before trying, given the 5-day half-life and time required for full clearance. Safety in patients with prior pancreatitis is unestablished, and severe gastroparesis is generally a reason to avoid the drug. Tirzepatide is not approved for type 1 diabetes or for pediatric use, although phase 3 pediatric trials in adolescent obesity (SURMOUNT-ADOLESCENTS) are ongoing.
Special populations
Renal impairment. No dose adjustment is required at any stage of chronic kidney disease, including dialysis-dependent patients, since metabolism does not depend on renal clearance [9]. Watch for dehydration-driven AKI in patients with reduced baseline GFR.
Hepatic impairment. No dose adjustment in mild, moderate, or severe hepatic impairment.
Older adults. Roughly 30 percent of trial participants were 65 or older. Efficacy and safety match younger adults, with somewhat higher rates of nausea and dehydration. Slower titration is reasonable for frail patients.
Bariatric surgery history. Tirzepatide can be used after sleeve gastrectomy or gastric bypass, often at lower doses, when weight regain occurs. Data are limited but accumulating in observational registries.
Compounded tirzepatide and the supply situation
Eli Lilly resolved the US tirzepatide shortage in late 2024, and the FDA confirmed in February 2025 that the molecule no longer met the criteria for compounding under section 503A or 503B of the Federal Food, Drug, and Cosmetic Act. After a wind-down period, compounding pharmacies and outsourcing facilities lost the ability to produce copies of tirzepatide. Patients who had been receiving compounded versions through telehealth platforms have transitioned either to branded Mounjaro or Zepbound through traditional pharmacy channels or to Lilly's direct-pay vials.
Compounded tirzepatide poses three concerns even when legal: ingredient variability (the active compound is not always pharmaceutical-grade tirzepatide and may contain salt forms or related peptides not approved for human use), dosing accuracy (patient-drawn injections from multidose vials are error-prone), and lack of regulatory oversight on sterility. Patients still considering non-FDA-approved sources should weigh those risks against the cost gap.
LillyDirect self-pay and access
Lilly's LillyDirect platform sells single-dose Zepbound vials to cash-paying patients without insurance for obesity drugs. As of 2026, vial pricing runs roughly 349 dollars for the 2.5 mg starter month, 499 dollars for 5 mg, 599 dollars for 7.5 mg, and 699 dollars for 10, 12.5, and 15 mg, all before any tax or shipping [13]. The vials require a prescription and are delivered through a contracted mail-order pharmacy. Patients draw the dose with a separate insulin syringe rather than using the prefilled autoinjector pen.
For patients with commercial insurance, the Mounjaro Savings Card and Zepbound Savings Card can drop monthly copays to as low as 25 dollars when the prescription is covered, and to several hundred dollars when it is not. Patients on Medicare, Medicaid, or other government programs are not eligible for these manufacturer cards under federal anti-kickback rules. Detailed cost and access guidance is in the dedicated cost and insurance reference.
Injection technique and minimizing local reactions
The single-dose autoinjector pen is designed for a button press without dose dialing. Patients pull off the gray base cap, place the clear base flat against the skin, and press the lock ring down until they hear a click and a second click after about 5 to 10 seconds. The pen is then lifted away. The vial presentation requires a separate insulin syringe, removal of the rubber stopper protector, drawing 0.5 mL with the needle inverted, and injecting at a 90-degree angle. Most injection-site irritation comes from injecting into a tense or recently shaved area, injecting cold drug straight from the fridge, or repeatedly using the same square inch of skin. Letting the pen reach room temperature for about 30 minutes before use, rotating sites in a four-quadrant abdomen pattern, and applying mild pressure rather than rubbing after the injection cuts most local discomfort.
Pipeline and future indications
Tirzepatide is being studied in cardiovascular outcomes (SURPASS-CVOT, comparing it head-to-head with dulaglutide in roughly 13,000 adults with type 2 diabetes and cardiovascular risk), pediatric obesity (SURMOUNT-ADOLESCENTS), MASH phase 3 (SYNERGY-NASH-2), chronic kidney disease in obesity, and heart failure phenotypes beyond HFpEF. Lilly is also developing retatrutide, a triple agonist that adds glucagon receptor activity to the GIP/GLP-1 backbone and produced about 24 percent weight loss at 48 weeks in phase 2, plus orforglipron, an oral non-peptide GLP-1 agonist that may compete on convenience.
Practical follow-up notes
Onset of appetite suppression is usually noticeable within the first 1 to 2 weeks. Average weight loss accumulates steadily through about month 18 on a stable maximum dose before plateauing. A1c lowering is faster, with most of the drop visible by week 24. Clinicians typically check A1c, weight, blood pressure, and a basic metabolic panel every 3 months during titration and every 6 months once on a stable dose. Persistent severe nausea, signs of pancreatitis, gallbladder symptoms, or unexplained mood changes warrant a treatment pause and reassessment.
Body composition and beyond-the-scale changes
Weight on the scale tells only part of the story. DXA substudies from the SURMOUNT program reported that roughly 75 percent of weight lost on tirzepatide was fat mass and 25 percent lean mass, a ratio similar to other significant weight loss interventions including bariatric surgery and diet alone at matched magnitudes. Visceral adipose tissue dropped disproportionately, and skeletal muscle index largely tracked the lean-to-fat ratio expected for the absolute weight change rather than showing accelerated muscle loss. Resistance exercise and adequate protein intake (1.2 to 1.6 g/kg of ideal body weight per day) help preserve lean mass during active weight loss, and most obesity medicine clinicians now build that counseling into the visit at the time of the first dose escalation.
Tirzepatide also drives reductions in liver fat, blood pressure (roughly 6 to 8 mmHg systolic at the 15 mg dose in pooled trials), triglycerides, fasting insulin, and high-sensitivity CRP that exceed what would be predicted by weight loss alone, suggesting some direct metabolic effects at the receptor level. Sleep quality improves, knee pain in osteoarthritis improves, and reflux symptoms generally improve as weight comes down, although a minority of patients experience worsened reflux from delayed gastric emptying.
Switching from semaglutide or other GLP-1 agonists
Patients moving from semaglutide, dulaglutide, or liraglutide to tirzepatide should wait one full dosing interval after the last GLP-1 dose, then start tirzepatide at 2.5 mg and titrate as a new patient. Skipping the 2.5 mg starter and jumping to a higher dose because the patient was already tolerating a different incretin is not endorsed by the label and tends to produce avoidable nausea. The two molecules clear at different rates and bind different receptor combinations, so prior tolerability does not translate cleanly. The reverse switch (tirzepatide to semaglutide) follows the same logic: wait 1 week after the last tirzepatide dose, then start semaglutide at 0.25 mg.
Clinicians who switch a patient because of payer coverage rather than clinical failure should warn the patient about a temporary period of reduced appetite suppression during the new titration. A bridging dose of stronger lifestyle support during weeks 1 through 8 of the new drug helps prevent backlash weight regain.
Plateaus, dose intensification, and stopping
Most patients reach a weight plateau after 12 to 18 months on a stable dose. Plateaus are not failures. They reflect the new homeostatic setpoint defended by the drug and lifestyle context. Options at plateau include accepting the current weight as the maintenance target, intensifying lifestyle measures (especially structured resistance training and protein-prioritized eating), increasing the dose if not already at 15 mg, switching to a more potent agent like retatrutide once approved, or, in a small subset, considering bariatric surgery as an additive layer.
Stopping tirzepatide remains a difficult conversation. SURMOUNT-4 quantified the regain risk: about two-thirds of weight loss returned within a year of discontinuation. The implication is that obesity behaves like other chronic diseases, where removing the treatment removes the response. Clinicians should frame tirzepatide as a long-term medication and budget cost and access accordingly. Patients planning a deliberate stop (pregnancy, surgery, cost change) should taper down to the lowest effective dose first, intensify lifestyle support, and plan a follow-up visit at 3 and 6 months to catch and react to regain early.
References
- U.S. Food and Drug Administration. FDA Approves Novel, Dual-Targeted Treatment for Type 2 Diabetes. FDA Press Announcement, 2022. fda.gov
- U.S. Food and Drug Administration. FDA Approves New Medication for Chronic Weight Management. FDA Press Announcement, 2023. fda.gov
- U.S. Food and Drug Administration. FDA Approves First Medication for Obstructive Sleep Apnea. FDA Press Announcement, December 2024. fda.gov
- Cleveland Clinic. Tirzepatide Injection. my.clevelandclinic.org, 2024. clevelandclinic.org
- Farzam K, Patel P. Tirzepatide. StatPearls, NCBI Bookshelf, 2024. ncbi.nlm.nih.gov
- National Library of Medicine. Tirzepatide Injection: MedlinePlus Drug Information. MedlinePlus, 2024. medlineplus.gov
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1). New England Journal of Medicine, 2022. nejm.org
- Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes (SURPASS-2). New England Journal of Medicine, 2021. nejm.org
- Wikipedia contributors. Tirzepatide. Wikipedia, 2026. en.wikipedia.org
- Aronne LJ, Horn DB, le Roux CW, et al. Tirzepatide as Compared with Semaglutide for the Treatment of Obesity (SURMOUNT-5). New England Journal of Medicine, 2025. nejm.org
- Malhotra A, Grunstein RR, Fietze I, et al. Tirzepatide for the Treatment of Obstructive Sleep Apnea and Obesity (SURMOUNT-OSA). New England Journal of Medicine, 2024. pubmed.ncbi.nlm.nih.gov
- American Diabetes Association. Latest Data From SURPASS Trials Demonstrate Tirzepatide Provided Blood Sugar Reductions and Weight Loss. diabetes.org, 2021. adameetingnews.org
- WeightWatchers Editorial Team. Zepbound vs Mounjaro: What Is the Difference?, 2025. weightwatchers.com
- Packer M, Zile MR, Kramer CM, et al. Tirzepatide for Heart Failure with Preserved Ejection Fraction and Obesity (SUMMIT). New England Journal of Medicine, 2024. pubmed.ncbi.nlm.nih.gov
- Loomba R, Hartman ML, Lawitz EJ, et al. Tirzepatide for Metabolic Dysfunction-Associated Steatohepatitis with Liver Fibrosis (SYNERGY-NASH). New England Journal of Medicine, 2024. pubmed.ncbi.nlm.nih.gov