What Is GLP-1?
Summary: GLP-1 is a gut hormone that tells the pancreas to release insulin, slows stomach emptying, and quiets appetite. The injectable and oral drugs people now call "GLP-1s" (Ozempic, Wegovy, Mounjaro, Zepbound, Rybelsus and others) are engineered copies of that hormone designed to last days instead of minutes.
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.
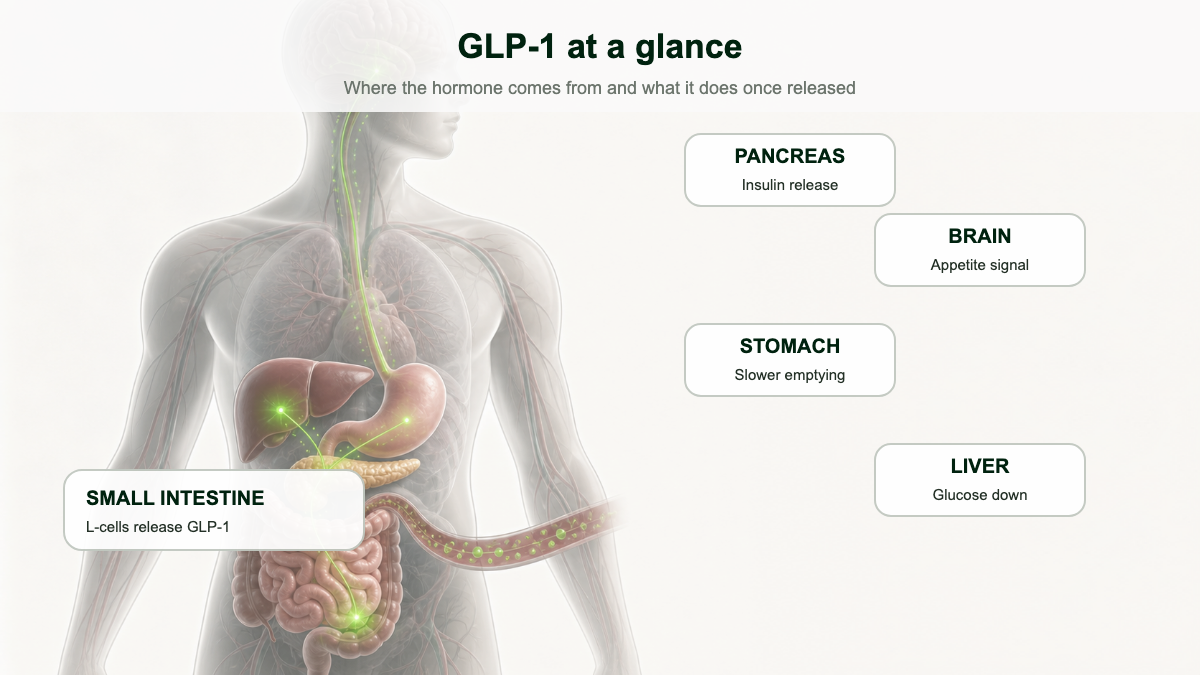
GLP-1 the hormone, in one paragraph
GLP-1 stands for glucagon-like peptide-1. It is a 30 or 31 amino acid peptide hormone made mainly by L cells in the lining of the small intestine and colon, with a smaller pool produced by neurons in the brainstem [1][2]. Specialized cells release it within minutes of food, especially carbohydrates and fats, hitting the bloodstream as nutrients arrive [1].
Once it is in circulation, GLP-1 does four useful things at once. It tells the pancreas to release insulin, but only when blood sugar is high. It blocks the opposing hormone glucagon. It slows the rate at which the stomach empties into the small intestine. And it acts on the hypothalamus to dial down hunger and bring on a sense of fullness [3][4].
Where the molecule comes from
GLP-1 is not synthesized as a finished hormone. It is carved out of a much larger precursor called proglucagon, the same gene product that, in pancreatic alpha cells, gets cut into glucagon. Tissue context decides which fragments survive. Alpha cells use a prohormone convertase called PCSK2 and end up releasing glucagon. Intestinal L cells and a small group of brainstem neurons use PCSK1 instead, and that enzyme yields GLP-1, GLP-2, glicentin, and oxyntomodulin in roughly equal molar amounts [5]. The two biologically active forms of GLP-1 in humans are GLP-1(7-37) and GLP-1(7-36)amide. More than 80 percent of what circulates in healthy adults is the amidated version [2][5].
L cells are not evenly distributed along the gut. Their density is sparse in the duodenum and rises through the jejunum and ileum, peaking in the colon [5]. Each cell is shaped a bit like a flask: a narrow apical tip pokes through the epithelium and tastes the contents of the lumen directly, while the basolateral side, where granules of GLP-1 are stored, sits next to capillaries and nerve endings. That architecture is why oral nutrients trigger a much bigger insulin response than the same nutrients given intravenously, the phenomenon explained later in this article as the incretin effect.
What sets GLP-1 release in motion
Glucose, fructose, long-chain fatty acids, di- and tri-peptides, and certain free amino acids all stimulate L cells, each through a different sensor. Glucose enters via SGLT1 and triggers depolarization through ATP-sensitive potassium channels, while fats activate G protein-coupled receptors such as GPR40 and GPR120 [5]. Bile acids in the distal small intestine and colon hit a receptor called TGR5 and add another layer of stimulation, which is part of why bariatric surgery, by rerouting bile flow, drives such large rises in postprandial GLP-1.
The vagus nerve coordinates the timing. Acetylcholine acts on muscarinic M1 and M2 receptors on L cells; gastrin-releasing peptide from enteric neurons enhances secretion further. In healthy volunteers, infusing atropine blunts the GLP-1 response to a meal, evidence that neural input is doing real work alongside the direct nutrient signal [5]. Hormones layer on top of that. Leptin activates hindbrain GLP-1 neurons. Ghrelin and cholecystokinin tweak central release. The system is redundant by design.
Why your own GLP-1 is not a drug
The natural hormone has a problem: it is gone almost as fast as it appears. An enzyme called dipeptidyl peptidase-4 (DPP-4) clips the active form apart, leaving a half-life of roughly two minutes in plasma. Only about 10 to 15 percent of secreted GLP-1 ever reaches the rest of the body intact [2][5].
That is fine for a meal-by-meal signal. It is useless as a medication. The whole story of the GLP-1 drug class is the story of chemists rebuilding the molecule so that DPP-4 cannot get a grip on it, and so the kidneys cannot flush it out in an afternoon.
Two distinct strategies emerged from that problem. The first is to inhibit DPP-4 itself, the basis of the gliptin drugs (sitagliptin, linagliptin, alogliptin) that prolong endogenous GLP-1 by about two- to three-fold but produce only modest glucose lowering and essentially no weight loss. The second strategy, the one that turned into Ozempic and Mounjaro, is to redesign the peptide so the enzyme cannot recognize it in the first place. The two approaches are sometimes confused in news coverage; they are pharmacologically unrelated despite both touching incretin biology.
What "incretin effect" means and why it matters in diabetes
If you swallow glucose, your pancreas releases far more insulin than if the same dose of glucose is dripped straight into a vein. The gap is called the incretin effect, and GLP-1 is one of the two hormones (the other is GIP, glucose-dependent insulinotropic polypeptide) that drives it [2]. In healthy adults the incretin effect accounts for roughly 50 to 70 percent of the insulin released after a meal [5].
In type 2 diabetes the incretin effect is blunted. People with the condition still respond to injected GLP-1, however, which is what made the hormone such an attractive drug target in the first place [2]. Because GLP-1's insulin signal is glucose-dependent, the pancreas mostly stops being prodded once blood sugar drops to normal, which keeps the risk of hypoglycemia much lower than with sulfonylureas or insulin alone [3].
GLP-1 vs GIP vs GLP-2: not interchangeable
The two human incretins look superficially similar but behave differently. GIP is 42 amino acids long, secreted by K cells in the upper small intestine, and has a half-life of about five minutes. GLP-1 is roughly 30 amino acids, secreted by L cells in the lower gut, and clears in about two [6]. In healthy people both stimulate insulin release with comparable potency. In type 2 diabetes the response to pharmacologic GIP is largely lost while the response to GLP-1 is preserved, a divergence that is still not fully explained but probably reflects a mix of GIP-receptor downregulation, accelerated degradation, and possibly receptor splicing differences [6]. GIP also tends to promote fat storage and bone formation; GLP-1 does neither. The fact that tirzepatide (Mounjaro, Zepbound) outperforms pure GLP-1 agonists by activating both receptors, despite GIP appearing useless on its own in diabetes, is one of the active research puzzles in the field.
GLP-2 is the sibling peptide cleaved out of the same proglucagon precursor inside L cells. It does not affect blood sugar or appetite at all. Instead it makes the intestinal lining grow. Teduglutide, a synthetic GLP-2 analogue, is FDA approved for short bowel syndrome to help patients absorb more nutrients orally and reduce dependence on intravenous nutrition. There is no clinically established GLP-3 in humans; some references use the label informally for other proglucagon fragments, but it has no defined receptor or therapeutic application.
From Gila monster spit to Ozempic: a quick history
Werner Creutzfeldt's group at Göttingen first described GLP-1 in 1979, and the proglucagon gene was cloned by Graeme Bell at Chiron in 1983, which made the precursor sequences readable for the first time [2]. Joel Habener's lab at Massachusetts General Hospital in Boston spent the mid-1980s working out which fragments of proglucagon were biologically active. Svetlana Mojsov, a peptide chemist in Habener's group, isolated the truncated GLP-1(7-37) form and demonstrated that it, not the full-length peptide, was the molecule that actually stimulated insulin release from human islets [2]. In parallel, Daniel Drucker, then a postdoc with Habener and now at Mount Sinai in Toronto, showed that GLP-1 acted directly on pancreatic beta cells. In Copenhagen, Jens Juul Holst's lab independently characterized the same insulinotropic activity and, later, the satiety effect [7].
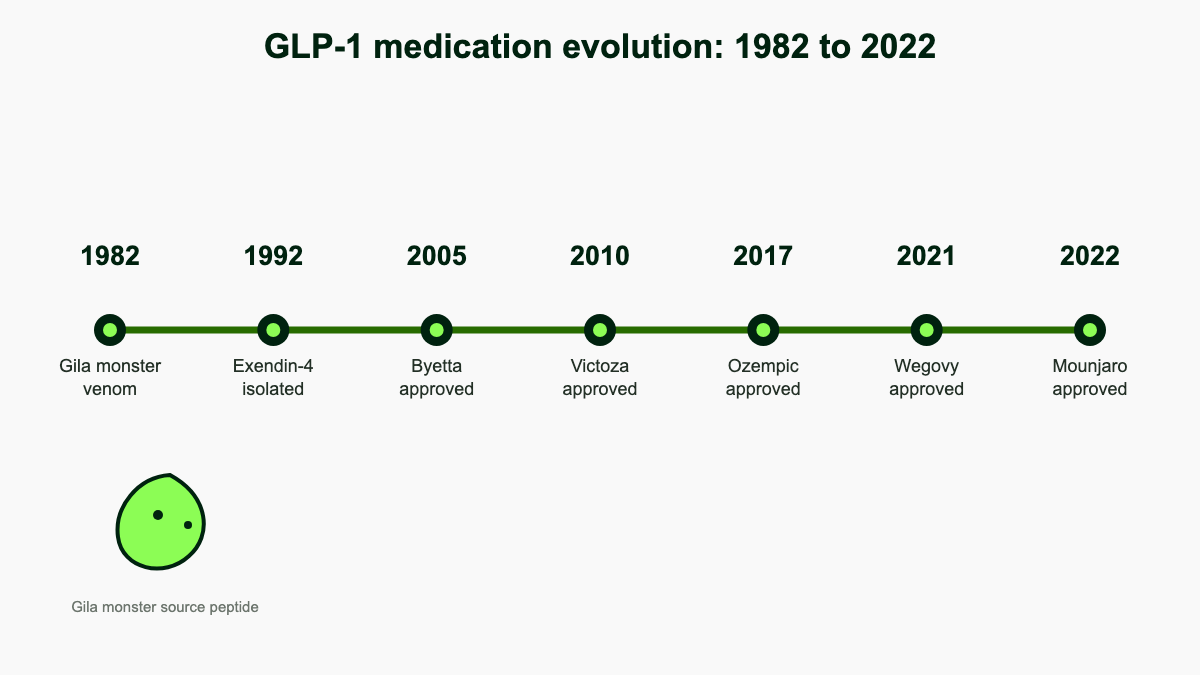
The Gila monster pivot
By the early 1990s the limitation was obvious: GLP-1 worked beautifully in infusion experiments but lasted minutes, which made it useless as an outpatient drug. The breakthrough came from John Eng, an endocrinologist at the Bronx VA Medical Center who was screening venoms for hormone-like activity. The Gila monster, Heloderma suspectum, lives in the Sonoran Desert and feeds only a few times a year. Eng reasoned that a lizard with such an erratic eating schedule might use unusual peptides to control its metabolism. In 1992 he isolated exendin-4 from its salivary secretions, a 39-amino acid peptide that activated the GLP-1 receptor and, crucially, was not a substrate for DPP-4 [3]. The patent went to the VA, the synthetic version was licensed to Amylin Pharmaceuticals, and Byetta (exenatide) reached the U.S. market in April 2005 as the first incretin-mimetic drug.
Albumin tethering and the road to weekly dosing
The next leap came from Novo Nordisk in Denmark. Lotte Bjerre Knudsen and her chemistry team kept the human GLP-1 backbone but attached a 16-carbon fatty acid side chain through a glutamic acid linker. The fatty acid bound non-covalently to albumin, the most abundant protein in blood, which protected the peptide from kidney filtration and slowed DPP-4 access. The result, liraglutide, was approved as Victoza for type 2 diabetes in 2010 and at a higher dose as Saxenda for obesity in 2014. Semaglutide pushed the same concept further by switching the linker chemistry and lengthening the fatty acid to 18 carbons, producing a half-life of roughly seven days and the first weekly GLP-1 (Ozempic, 2017; Wegovy at the 2.4 mg obesity dose, 2021). Eli Lilly's tirzepatide, approved as Mounjaro in 2022 and Zepbound in 2023, used a similar fatty acid-anchor strategy on a peptide engineered to hit both the GLP-1 and GIP receptors.
Timeline at a glance
- 1979 Creutzfeldt names the incretin concept; GLP-1 inferred from proglucagon.
- 1983 Bell clones proglucagon; full sequence available.
- 1986-1987 Mojsov, Habener, Holst, Drucker show GLP-1(7-37) is the insulinotropic fragment.
- 1992 Eng isolates exendin-4 from Gila monster venom.
- April 2005 Byetta (exenatide) approved by FDA for type 2 diabetes.
- January 2010 Victoza (liraglutide) approved.
- January 2012 Bydureon (weekly exenatide) approved.
- September 2014 Trulicity (dulaglutide) approved; Saxenda (high-dose liraglutide) approved for obesity.
- July 2016 Adlyxin (lixisenatide) approved.
- December 2017 Ozempic (injectable semaglutide) approved.
- September 2019 Rybelsus (oral semaglutide) approved, the first oral GLP-1.
- June 2021 Wegovy (semaglutide 2.4 mg) approved for chronic weight management.
- May 2022 Mounjaro (tirzepatide) approved for type 2 diabetes.
- November 2023 Zepbound (tirzepatide) approved for chronic weight management.
- March 2024 Wegovy gains an indication for cardiovascular risk reduction in adults with overweight or obesity and established cardiovascular disease.
- December 2024 Zepbound approved for moderate-to-severe obstructive sleep apnea in adults with obesity, the first drug ever approved for that condition.
What a "GLP-1 receptor agonist" actually is
An agonist is a molecule that binds to a receptor and turns it on the same way the natural ligand does [3]. GLP-1 receptor agonists (often shortened to GLP-1 RAs or just "GLP-1s") attach to the same GLP-1 receptors on pancreatic beta cells, gastric muscle, and brain regions that the hormone normally hits, but they linger long enough to be dosed daily, weekly, or in one case as an oral tablet.
Inside the receptor
The GLP-1 receptor (gene name GLP1R, on chromosome 6p21.2) is a class B G protein-coupled receptor of the secretin family. It has the canonical seven-helix transmembrane core plus a large extracellular N-terminal domain that grips the C-terminal half of GLP-1 first, in a "two-domain" binding mode characteristic of class B receptors [8]. Once GLP-1 or an agonist is locked in, the receptor couples primarily to the Gαs subunit, activates adenylate cyclase, and lifts intracellular cyclic AMP. cAMP turns on protein kinase A (PKA) and the cAMP-responsive guanine nucleotide exchange factor Epac2, both of which converge on insulin granule release in beta cells [5][8]. A parallel arm of signaling runs through β-arrestin recruitment, which is independent of G proteins and influences receptor internalization, recycling, and downstream gene transcription.
That second pathway matters because not every agonist activates both arms in the same proportion. Drugs that favor cAMP signaling and avoid heavy β-arrestin recruitment seem to produce more sustained insulin secretion with less receptor desensitization, an idea called "biased agonism" that is now actively shaping next-generation drug design [8]. Tirzepatide, for example, behaves as an "imbalanced" agonist at the GLP-1 receptor, with relatively low β-arrestin engagement compared with native GLP-1.
The receptor is almost everywhere useful
GLP-1 receptors are dense on pancreatic beta cells, on smooth muscle and neurons of the stomach, and on neurons in the hypothalamic arcuate nucleus and paraventricular nucleus that govern feeding [5]. They are also present in the heart (atrial myocytes and the sinoatrial node), kidney (proximal tubule and afferent arteriole), lung, and liver. Within the brain, expression extends to the area postrema, nucleus tractus solitarii, ventral tegmental area, and nucleus accumbens, the last two being core nodes of the dopamine reward circuit. That distribution is why GLP-1 drugs can lower blood pressure modestly, increase natriuresis, slow heart rate slightly, and, plausibly, dampen reward-driven cravings for food, alcohol, and nicotine [4].
The current U.S. lineup
By generic name and brand:
- Exenatide (Byetta, twice daily; Bydureon BCise, weekly) for type 2 diabetes [3].
- Liraglutide (Victoza, daily) for type 2 diabetes; (Saxenda, daily, higher dose) for chronic weight management [3].
- Lixisenatide (Adlyxin, daily) for type 2 diabetes [3].
- Dulaglutide (Trulicity, weekly) for type 2 diabetes [3].
- Semaglutide injection (Ozempic, weekly) for type 2 diabetes; (Wegovy, weekly, 2.4 mg) for chronic weight management; oral tablet (Rybelsus, daily) for type 2 diabetes [3][4].
- Tirzepatide (Mounjaro, weekly) for type 2 diabetes; (Zepbound, weekly) for chronic weight management. Tirzepatide is technically a dual agonist that hits both the GLP-1 and GIP receptors, but it is usually grouped with the GLP-1 class in clinical conversation [3].
What these medications are approved to treat
Type 2 diabetes
Every drug in the class above is approved as an add-on for adults with type 2 diabetes whose blood sugar is not controlled by diet, exercise, or other oral medications. They typically lower hemoglobin A1c by about 1 to 2 percentage points and produce modest weight loss as a side effect. In the SUSTAIN trial program, semaglutide reduced A1c by 1.4 to 1.8 percentage points depending on dose; in the SURPASS trials, tirzepatide dropped A1c by up to 2.4 points at the 15 mg dose, the largest reduction ever shown by a non-insulin diabetes drug.
Chronic weight management
Three products are FDA-approved specifically for obesity, not diabetes: Saxenda (liraglutide 3.0 mg), Wegovy (semaglutide 2.4 mg), and Zepbound (tirzepatide). They are indicated for adults with a body mass index of 30 or higher, or 27 or higher with at least one weight-related condition such as hypertension or sleep apnea. Average weight loss at 68 weeks is about 6 percent for liraglutide, 15 percent for semaglutide (STEP 1), and 21 percent for tirzepatide at the 15 mg dose (SURMOUNT-1). Roughly a third of patients on tirzepatide lose more than 25 percent of starting body weight, results that previously required bariatric surgery to achieve.
Cardiovascular disease
The first hint that GLP-1 drugs did more than control glucose came from the LEADER trial in 2016, which showed that liraglutide cut major cardiovascular events by 13 percent in adults with type 2 diabetes and high cardiovascular risk. SUSTAIN-6 found similar benefit for semaglutide. The SELECT trial, published in 2023, then made the case in non-diabetic patients: in 17,604 adults with overweight or obesity and established cardiovascular disease, weekly semaglutide 2.4 mg reduced the composite of cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke by 20 percent over a median of 40 months. The FDA added that indication to Wegovy in March 2024 [4].
Kidney, liver, and sleep
The FLOW trial, halted early for benefit, showed semaglutide slowed the progression of chronic kidney disease in patients with type 2 diabetes by 24 percent. In MASH (metabolic dysfunction-associated steatohepatitis, formerly NASH), semaglutide trials have shown improvement in steatosis and inflammation, with submissions for that indication anticipated. Zepbound's approval in December 2024 for moderate-to-severe obstructive sleep apnea, based on the SURMOUNT-OSA trial showing a roughly two-thirds reduction in apnea-hypopnea index, was the first drug ever approved for OSA, a condition that had only mechanical treatments before.
Emerging uses being studied
Active or recently completed trials are looking at GLP-1 drugs in heart failure with preserved ejection fraction (semaglutide showed benefit in STEP-HFpEF), Alzheimer's disease (the EVOKE program for semaglutide is enrolling), Parkinson's disease, alcohol use disorder, opioid use disorder, polycystic ovary syndrome, and osteoarthritis pain through weight loss. None of these are FDA-approved indications today, but the breadth of investigation reflects how broadly the GLP-1 receptor is expressed [4].
Side effects you actually hear about
The dominant side effects across the class are gastrointestinal: nausea, vomiting, diarrhea, constipation, stomach pain, and early satiety [3][4]. In the STEP and SURMOUNT trials nausea was reported by 40 to 45 percent of patients on semaglutide and tirzepatide compared with about 16 percent on placebo, vomiting by roughly a quarter, diarrhea by a fifth. They are usually worst in the first few weeks and after each dose increase, which is why prescribers titrate up over months rather than starting at the full therapeutic level. Roughly 5 to 10 percent of patients discontinue treatment because of GI symptoms in the trials [9].
The serious-but-rare warnings on every U.S. label include acute pancreatitis (incidence around 0.1 to 0.2 percent per year, similar to background in the diabetes population) and a boxed warning about thyroid C-cell tumors based on rodent studies; the human relevance is uncertain, but the drugs are not used in people with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia type 2 [4]. Gallbladder problems are more common in obesity trials, partly because rapid weight loss itself raises gallstone risk. Loss of muscle mass alongside fat is real and dose-dependent, which is why most clinicians pair these drugs with adequate protein intake and resistance training. The UK MHRA is investigating very rare reports of non-arteritic anterior ischemic optic neuropathy (NAION), a sudden vision-loss condition, in semaglutide users; the absolute risk, if any, appears small [10].
Anesthesia, surgery, and other practical points
Because GLP-1 drugs slow gastric emptying for days, patients can show up to elective surgery with food still in the stomach despite an overnight fast, raising the risk of pulmonary aspiration during anesthesia. The American Society of Anesthesiologists and several international bodies now recommend pausing GLP-1s before procedures, generally one week for weekly drugs and one day for daily ones. The drugs can also blunt the effectiveness of oral contraceptive pills around tirzepatide dose increases, which is why MHRA guidance asks women on Mounjaro to use a barrier method for four weeks after starting and after each titration step [10]. Pregnancy is a hard contraindication; recommended washout before conception is two months for semaglutide, one month for tirzepatide, and immediately for liraglutide [10]. Breastfeeding is also discouraged because of inadequate human safety data.
Drug interactions are mostly about timing rather than chemistry. The slowed stomach can delay or reduce absorption of co-administered oral medications, and adding a GLP-1 to a sulfonylurea or basal insulin substantially raises hypoglycemia risk; most clinicians cut the sulfonylurea or insulin dose by about 25 percent at initiation [9]. There is no rationale for combining a GLP-1 receptor agonist with a DPP-4 inhibitor, since the latter just prolongs endogenous GLP-1 that is already being out-signalled by the agonist; insurers usually deny that combination.
Common misconceptions about GLP-1
"It's just an appetite suppressant"
The appetite effect is real but partial. Half the weight loss with semaglutide comes from reduced food intake; the rest comes from a combination of altered food preferences (fatty and sweet foods become less rewarding), slower gastric emptying that prolongs satiety between meals, and small but measurable changes in resting energy expenditure. Patients often describe quieting of "food noise," the constant low-level mental presence of what to eat next. That symptom relief is one of the more striking subjective changes that older appetite suppressants like phentermine never produced.
"Ozempic is a weight-loss drug"
Ozempic is approved only for type 2 diabetes. Wegovy is the same molecule (semaglutide) approved at the higher 2.4 mg weekly dose for chronic weight management. Insurance coverage and prescribing rules differ between them, which is why the two brand names exist for one drug.
"GLP-1s are addictive"
GLP-1 receptor agonists do not act on opioid, dopamine, or benzodiazepine systems directly, do not produce a high, and have no documented withdrawal syndrome. What does happen, predictably, is that appetite returns and weight tends to come back when treatment is stopped, because obesity behaves more like hypertension than like a finite course of antibiotics. The STEP-1 extension showed that two-thirds of weight lost on semaglutide was regained within a year of stopping the drug.
"They are dangerous in the long term"
Liraglutide has been on the market since 2010, exenatide since 2005. The accumulated patient-years now run into the millions, with no signal of new long-term harms beyond those listed on the label. Long-term cardiovascular and renal data, in fact, point the other direction: less risk, not more.
What "natural" GLP-1 boosters can and cannot do
You will see claims that protein, fiber, fermented foods, berberine, apple cider vinegar, or specific supplements "boost GLP-1." Eating does raise GLP-1, briefly, because that is the hormone's job. The increase is on the order of a few picomoles per liter for an hour or two, then the DPP-4 enzyme grinds it back down [5]. That is not in the same neighborhood as the steady, multi-day exposure produced by a weekly semaglutide or tirzepatide injection.
Some specifics are worth getting right. Whey protein, soluble fiber (especially fermentable fibers like inulin and beta-glucan), monounsaturated fats from olive oil, and short-chain fatty acids produced by gut bacteria from fiber fermentation all increase postprandial GLP-1 in human studies, often by 30 to 50 percent above a low-fiber control meal. The clinical translation is modest. None of these has been shown in a randomized trial to produce the 10 to 20 percent body-weight reduction that the drugs deliver. Berberine, sometimes marketed as "nature's Ozempic," is a plant alkaloid that activates AMPK and lowers blood glucose by a mechanism unrelated to GLP-1; the meme is catchy and pharmacologically inaccurate. Apple cider vinegar slows gastric emptying slightly through an acid effect, again unrelated to incretin signaling. Bariatric surgery is the one non-drug intervention that does drive sustained, large rises in postprandial GLP-1 (often five- to ten-fold), which is part of why gastric bypass produces durable weight loss.
Diet matters for plenty of reasons. The pharmacology of food and the pharmacology of these drugs are not interchangeable.
The drug pipeline beyond Ozempic and Mounjaro
The current GLP-1 list is the opening round. The pipeline is dense [11].
Oral small-molecule GLP-1 agonists
Until now, every GLP-1 drug has been a peptide, which is hard to deliver as a pill because stomach acid and proteases destroy it. Rybelsus solves the problem with a permeation enhancer called SNAC, but bioavailability is still under 1 percent and the pill must be taken on an empty stomach with strict water rules. Eli Lilly's orforglipron is a non-peptide small molecule that activates the GLP-1 receptor directly and survives oral absorption without those constraints. Phase 3 ATTAIN-1 and ACHIEVE-1 trials reported in 2025 showed roughly 11 percent weight loss and A1c reductions comparable to injectable semaglutide. Filings are expected through 2026 and 2027 [11]. Pfizer's danuglipron has stumbled with liver enzyme elevations and cardiovascular safety concerns.
Dual and triple agonists
Tirzepatide proved that adding GIP activity to GLP-1 boosted efficacy. Eli Lilly's retatrutide goes further by hitting GLP-1, GIP, and the glucagon receptor at once. Glucagon agonism, paradoxically, increases energy expenditure rather than blood sugar in this context. In phase 2 trials retatrutide produced 24 percent weight loss at 48 weeks at the highest dose, the largest figure ever recorded for a single weight-loss drug. Phase 3 data and an FDA filing are expected in 2026 to 2027 [11]. Boehringer Ingelheim and Zealand Pharma's survodutide is a GLP-1/glucagon dual agonist showing strong activity in MASH liver disease. Innovent's mazdutide, another GLP-1/glucagon, is approved in China.
Amylin and combination peptides
Cagrilintide is an analogue of amylin, a beta-cell peptide that complements GLP-1 by acting on hindbrain satiety circuits. Novo Nordisk's CagriSema combines cagrilintide and semaglutide in one weekly injection; phase 3 data showed weight loss in the 20 percent range, putting it close to tirzepatide territory. Filings are anticipated in late 2026 [11]. Amgen's MariTide (maridebart cafraglutide) is a GLP-1-active monoclonal antibody peptide conjugate dosed monthly rather than weekly.
Why the patent cliff matters
Semaglutide's core composition-of-matter patents in the United States expire in 2032, with some peripheral patents extending into 2033 and 2034. Generic semaglutide is unlikely to reach U.S. pharmacies before then, although other markets (notably India, Brazil, and parts of Latin America) will see generic versions sooner because of different patent terms. Liraglutide is already off-patent in the U.S. as of 2024, with the first generics launched in 2025. The combination of patent expiry and oral small-molecule entrants is expected to drag prices down sharply in the late 2020s.
The bigger picture
GLP-1 is a small molecule with an outsized resume. As a hormone it tells the pancreas to release insulin only when needed, blocks glucagon, slows gastric emptying, and quiets hunger circuits in the hypothalamus. As a drug class, the engineered analogues do all of that for a week at a time, and along the way reduce major cardiovascular events, slow kidney decline, ease sleep apnea, and treat obesity with results that used to require surgery. The story is a 45-year arc from a venomous lizard and an obscure proglucagon fragment to one of the largest pharmaceutical categories in history. The pipeline points to oral pills, triple agonists, and monthly antibody-peptide conjugates that will keep reshaping the field through the rest of this decade.
References
- Cleveland Clinic. GLP-1 Agonists: What They Are, How They Work & Side Effects. Cleveland Clinic, 2024. my.clevelandclinic.org
- Wikipedia contributors. Glucagon-like peptide-1. Wikipedia, 2025. en.wikipedia.org
- Drugs.com. What are GLP-1 medications and how do they work?. Drugs.com, 2024. drugs.com
- MedlinePlus. Semaglutide Injection. U.S. National Library of Medicine, 2024. medlineplus.gov
- Müller TD, Finan B, Bloom SR, et al. Glucagon-like peptide 1 (GLP-1). PMC, 2019. pmc.ncbi.nlm.nih.gov
- Nauck MA, Meier JJ. GIP and GLP-1, the two incretin hormones: similarities and differences. PMC, 2014. pmc.ncbi.nlm.nih.gov
- Drucker DJ, Holst JJ. The GLP-1 journey: from discovery science to therapeutic impact. PMC, 2024. pmc.ncbi.nlm.nih.gov
- Zhao F, Zhou Q, Cong Z, et al. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Nature Signal Transduction and Targeted Therapy, 2024. nature.com
- StatPearls. Glucagon-Like Peptide-1 Receptor Agonists. NCBI Bookshelf, 2024. ncbi.nlm.nih.gov
- Medicines and Healthcare products Regulatory Agency. GLP-1 medicines for weight loss and diabetes: what you need to know. UK Government, 2025. gov.uk
- Prime Therapeutics. GLP-1 Pipeline Update: February 2026. Prime Therapeutics, 2026. primetherapeutics.com