GLP-1 Side Effects & Risks
Summary: Most people on a GLP-1 get gastrointestinal side effects (nausea, vomiting, diarrhea, constipation) that peak during dose escalation and usually settle within a few weeks. The serious risks are real but uncommon: pancreatitis, gallbladder disease, gastroparesis, a rodent-derived thyroid C-cell tumor warning, and acute kidney injury from dehydration.
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.

How to read this page
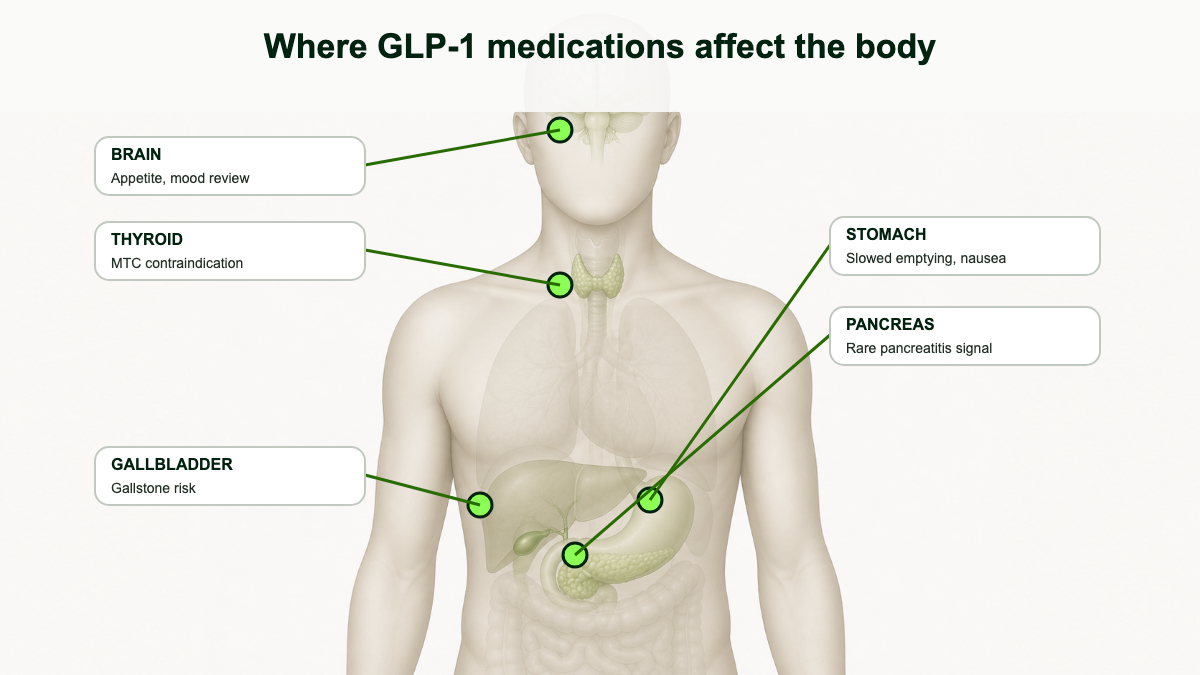
Side effects from GLP-1 receptor agonists fall into a small number of buckets, and grouping them by frequency makes the picture less alarming than scrolling through a label cold. The very common effects (more than 1 in 10 patients) are almost entirely gastrointestinal and dose-dependent. The common effects (1% to 10%) include injection-site reactions, fatigue, dizziness, burping, and hair shedding. The uncommon-but-clinically-important effects (under 1% but worth knowing about) include acute pancreatitis, gallbladder disease, severe gastroparesis, acute kidney injury from dehydration, and worsening of pre-existing diabetic retinopathy. The rare warning-level events are the boxed thyroid C-cell tumor warning (rodent data), pulmonary aspiration during anesthesia, and a now-removed suicidal ideation signal that did not survive formal review. [1][2][3]
Where this page cites a specific frequency, it is taken from a published FDA label or a pivotal trial. Real-world rates are usually lower than trial rates because trials titrate aggressively to test the maximum dose, while clinical practice slows down for tolerability. The numbers below are upper bounds, not what most people will actually feel.
The most common side effects: gastrointestinal
GI symptoms are the dominant side-effect signal across every GLP-1 receptor agonist on the market. In the STEP-1 trial of semaglutide 2.4 mg (Wegovy) for weight loss, gastrointestinal disorders affected 74.2% of patients on semaglutide versus 47.9% on placebo over 68 weeks. [1] The breakdown from STEP-1 looked roughly like this for semaglutide vs placebo:
- Nausea: about 44% vs 18%
- Diarrhea: about 32% vs 16%
- Vomiting: about 25% vs 7%
- Constipation: about 23% vs 10%
- Abdominal pain: about 20% vs 10%
Tirzepatide (Mounjaro, Zepbound) shows a similar pattern, with nausea reported in 12% to 18% of patients across the 5, 10, and 15 mg doses in SURMOUNT-1, and diarrhea in 12% to 17%. [2] Ozempic at the 1 mg dose produces nausea in roughly 1 in 5 patients and vomiting in about 1 in 10. [3] The Ozempic label lists nausea, vomiting, diarrhea, abdominal pain, and constipation as the most common adverse reactions reported in 5% or more of patients across all dose strengths. [3]
The good news: most events are mild to moderate, peak during titration when the dose is being increased, and fade within 4 to 8 weeks at a stable dose. About 7% of STEP-1 participants discontinued semaglutide because of adverse events, mainly GI, compared with 3.1% on placebo. [1] In the Zepbound trials, 3.3% of patients on the 10 mg dose and 4.3% on 15 mg discontinued for GI reasons compared with 0.5% on placebo. [4] If symptoms are severe or do not improve, the dose can be held or reduced rather than abandoning the drug entirely.
Why GI symptoms happen: GLP-1 (and the dual GLP-1/GIP agonist tirzepatide) slow gastric emptying. Food sits in the stomach longer, which is part of how the drug produces fullness, but the same mechanism produces nausea and reflux when too much food, or the wrong kind of food, is eaten. The slowed transit also contributes to constipation lower in the gut, while parallel effects on intestinal motility can drive diarrhea in others. Tirzepatide has a stronger effect on gastric emptying than pure GLP-1 agonists, which is one reason eructation (burping) is more prominent on it. [4]
Bottom line: expect some GI upset during the first weeks at each new dose. Slow titration, smaller meals, and avoiding fatty or fried food usually keep it manageable.
Drug-by-drug differences in side-effect profile
The class shares a common GI signature, but each molecule has quirks worth knowing about before picking one.
- Semaglutide (Ozempic, Wegovy, Rybelsus): the workhorse profile. Nausea is the leading complaint; diarrhea and constipation roughly tie for second. The oral form (Rybelsus) requires a fasting window, which itself produces a slightly different GI experience because peak drug exposure is more abrupt. [3][5]
- Tirzepatide (Mounjaro, Zepbound): slightly higher rates of eructation (burping) and dyspepsia than semaglutide, attributed to its dual GIP/GLP-1 mechanism and stronger effect on gastric emptying. The Zepbound label reports eructation in roughly 4% to 5% of patients across the 5, 10, and 15 mg doses, versus around 0.2% on placebo. [4] Sulfur-flavored burps are a frequent informal complaint and reflect food sitting longer in the stomach rather than a unique drug toxicity.
- Liraglutide (Victoza, Saxenda): daily injection, longer track record, slightly higher gallbladder signal in pooled analyses, possibly because earlier liraglutide weight-loss trials achieved meaningful but slower weight loss with chronic GI exposure. [6]
- Dulaglutide (Trulicity): weekly, similar nausea profile, less weight-loss data but a strong cardiovascular outcomes record in REWIND.
- Exenatide (Byetta, Bydureon): the original twice-daily exenatide produced more nausea than the weekly Bydureon depot, and nodules at the injection site are a known idiosyncrasy of the depot formulation.
None of these differences are large enough to make one drug categorically "safer" than another. Switching molecules within the class is a reasonable strategy when one specific side effect (often nausea or burping) is intolerable.
Injection site reactions and other mild effects
Local injection site reactions (redness, itching, mild swelling, induration) are reported in roughly 1.4% of Wegovy users in adult trials versus 1.0% on placebo, and at similar low rates with Ozempic. [3][7] Tirzepatide trials reported injection-site reactions in about 8.0% of Zepbound patients versus 1.8% on placebo, somewhat higher than the semaglutide products but still mild in nearly all cases. [4] They almost always resolve on their own. Rotating injection sites between abdomen, thigh, and upper arm reduces the chance of a hard lump, bruise, or persistent pruritus.
Other low-grade complaints that show up in trials and post-marketing reports include:
- Headache and fatigue, especially during the first weeks at each new dose
- Dizziness, often related to dehydration or rapid postural change
- Burping (eructation), flatulence, bloating, and acid reflux
- Altered taste (metallic, bitter, or simply blunted)
- Hair shedding, typically tied to rapid weight loss rather than the drug itself
- A modest resting heart rate increase of 2 to 4 beats per minute on average [3]
- Mild elevations in serum amylase and lipase without clinical pancreatitis [6]
Bottom line: these are nuisance effects, not danger signals, and most resolve once weight loss slows or the dose stabilizes.
Hair loss and alopecia: how much is from the drug
Hair shedding has become one of the most discussed cosmetic side effects of GLP-1 therapy. The Wegovy label reports alopecia in 3.5% of treated adults versus 1.0% on placebo. [7] The numbers climb with magnitude of weight loss: in the higher-dose oral semaglutide weight-loss trials, hair loss was reported in roughly 7% of patients on 50 mg/day versus 3% of controls, and people who lost 20% or more of their body weight reported it more often than those who lost less. [8]
The pattern in dermatology clinics is consistent with telogen effluvium, a stress-related diffuse shedding that tends to start 2 to 4 months after a metabolic stressor (rapid weight loss, surgery, illness, childbirth) and recovers spontaneously over 6 to 12 months as the hair cycle resets. [8] In other words, GLP-1s appear to trigger hair loss the same way any rapid weight loss does, not by acting on hair follicles directly. Practical mitigation:
- Slow the rate of weight loss if possible (longer titration, lower target dose)
- Hit a daily protein target of 1.2 to 1.6 g per kg of body weight
- Check ferritin, vitamin D, and zinc, which often run low during rapid weight loss
- Avoid crash-dieting on top of the drug
- Wait it out: most cases self-resolve once weight is stable
Bottom line: the shedding is real but typically transient and tied to weight-loss velocity rather than direct drug toxicity.
"Ozempic face" and lean mass loss
"Ozempic face" describes the hollowed cheeks, deeper folds, and looser skin some people notice after rapid weight loss on a GLP-1. The drug itself is not pulling fat out of the face; what is happening is general subcutaneous fat loss, including from facial fat pads, plus reduced skin elasticity that becomes visible when weight comes off fast. [9] The same phenomenon shows up after bariatric surgery and aggressive dieting. The cosmetic visibility is concentrated in patients over 40 with already reduced collagen content, which is part of why the look is more dramatic in older patients losing 15% or more of body weight.
A related and arguably more important concern is lean mass loss. The STEP-1 DXA substudy showed that total lean body mass fell by about 9.7% from baseline at week 68, while total fat mass fell by 19.3%. The proportion of lean tissue actually rose by 3.0 percentage points relative to fat, but absolute muscle still came off. [10] The SURMOUNT-1 DXA substudy of tirzepatide reported a 21.3% reduction in body weight, with fat mass down 33.9% and lean mass down 10.9% over 72 weeks. About 74% of the weight lost was fat and 26% was lean, a ratio that mirrored the placebo arm. [11]
That ratio matters: roughly a quarter of weight lost on a GLP-1, without dedicated resistance training and adequate protein, comes from lean tissue. This is not unique to GLP-1s, but the speed and magnitude of weight loss on semaglutide and tirzepatide make the issue more visible. Mitigation is straightforward in principle:
- Aim for 1.2 to 1.6 g of protein per kg of body weight per day
- Do progressive resistance training 2 to 3 times per week
- Avoid skipping meals despite suppressed appetite
- Discuss creatine and vitamin D with a clinician if intake is low
- Get a baseline DXA or bioimpedance reading before starting if available, and recheck after 6 to 12 months
Bottom line: the cosmetic and muscle-loss effects are consequences of rapid weight loss, not direct drug toxicity. Slower titration, protein, and lifting blunt them.
Pancreatitis: real but uncommon
Acute pancreatitis appears in the warnings section of every GLP-1 label. The absolute risk is small. In the LEADER cardiovascular outcomes trial of liraglutide, acute pancreatitis occurred in 0.4% of treated patients versus 0.5% of placebo, suggesting little or no excess at standard diabetes doses. [6] At higher weight-loss doses and in younger or female populations, postmarketing data have produced a stronger signal. A 2023 JAMA cohort study by Sodhi and colleagues compared GLP-1 users to bupropion-naltrexone users for weight loss and found an adjusted hazard ratio of 9.09 (95% CI 1.25 to 66.00) for pancreatitis, 3.67 (1.15 to 11.90) for gastroparesis, and 4.22 (1.02 to 17.40) for bowel obstruction. [12] Confidence intervals are wide and event counts low, but the signal is consistent enough that clinicians are told to discontinue the drug if pancreatitis is suspected and to avoid restarting it in patients with prior pancreatitis.
The symptom that warrants a same-day call is severe upper-abdominal pain, often radiating through to the back, frequently accompanied by persistent vomiting. Tenderness, fever, and inability to keep fluids down increase the urgency. Most patients who present with these features and stop the drug recover with supportive care; ongoing GLP-1 use after a clear pancreatitis episode is not advised in current labeling. [3][7]
Gallbladder and biliary disease
Gallbladder events (gallstones, cholecystitis, biliary colic) are reported in roughly 2.6% of Wegovy patients versus 1.2% on placebo in STEP-1. [1] A meta-analysis pooling 76 randomized controlled trials found a 37% higher relative risk of gallbladder or biliary disease with GLP-1 receptor agonists compared with controls, with the strongest signals for cholelithiasis (RR 1.27), cholecystitis (RR 1.36), and biliary disease overall (RR 1.55). The risk was greater at the higher doses used for weight loss and with longer treatment duration. [13]
Mechanism is partly the drugs and partly the weight loss itself. Rapid loss of more than 1.5% of body weight per week is a long-recognized risk factor for gallstone formation because of biliary cholesterol supersaturation, and GLP-1 agonists also reduce gallbladder contractility and emptying. Patients lose weight quickly on Wegovy and tirzepatide, so the two effects stack.
Practical signals to call about: right-upper-quadrant pain (especially after a fatty meal), jaundice, dark urine or pale stools, fever with abdominal pain. Most cases that progress to surgery are managed straightforwardly with cholecystectomy, and the GLP-1 typically does not need to be permanently stopped unless episodes recur.
Gastroparesis, ileus, and the lawsuit context
Severely delayed gastric emptying, sometimes labeled "stomach paralysis" in lay coverage, has driven both lawsuits and anesthesia guidance. The biology is straightforward: every GLP-1 receptor agonist slows gastric emptying as part of its mechanism, and in a small minority of patients the slowing becomes prolonged and clinically significant. Symptoms to flag are early satiety, bloating that lingers many hours after eating, vomiting undigested food, and persistent upper-abdominal pain.
The Sodhi cohort study estimated a roughly 3.7-fold higher risk of gastroparesis with GLP-1 use compared with bupropion-naltrexone, although the absolute incidence remained low. [12] In February 2024, the U.S. Judicial Panel on Multidistrict Litigation consolidated lawsuits against the GLP-1 manufacturers into MDL 3094 in the Eastern District of Pennsylvania. The MDL had grown to roughly 3,600 pending cases by mid-2026, alleging inadequate warnings about severe gastroparesis, intestinal obstruction, ileus, and (in a separate cluster) NAION-related vision loss. [14] The litigation does not by itself establish causation; it does reflect that the post-marketing GI signal is large enough to draw legal attention.
For the individual patient, the practical takeaway is unchanged from before the MDL: stop the drug if vomiting persists more than 24 hours, if abdominal pain is severe, or if gastroparesis-style fullness lasts beyond what dose titration can explain. Imaging and gastric emptying studies are available if symptoms continue after stopping.
Anesthesia and surgery: updated 2024 guidance
Because GLP-1s slow gastric emptying, residual stomach contents at the time of induction raise the risk of pulmonary aspiration. The American Society of Anesthesiologists released initial guidance in June 2023 recommending that weekly GLP-1s be held for one week and daily formulations for one day before elective procedures. [15] In October 2024, ASA and a multi-society panel (American Gastroenterological Association, American Society for Metabolic and Bariatric Surgery, IPSCOPO, and SAGES) reversed that blanket pause and issued more nuanced guidance: most patients can continue their GLP-1 perioperatively, with an individualized risk assessment based on shared decision-making between the patient, prescriber, and anesthesia team. [16]
Strategies the 2024 guidance endorses for reducing aspiration risk without stopping the drug include:
- A 24-hour clear-liquid diet before the procedure
- Point-of-care gastric ultrasound to check for retained contents in higher-risk patients
- Adjusting anesthesia plan (rapid sequence induction, head-up positioning) when stomach content is uncertain
- Identifying patients with bowel dysmotility, prior gastroparesis, or Parkinson's disease, who carry baseline higher risk
For endoscopy specifically, the same principles apply: extended fasting, liquid diet, and case-by-case decision rather than universal drug pause. Tell every surgeon, gastroenterologist, and anesthesiologist about your GLP-1 well before the procedure.
Thyroid C-cell tumor warning
Every GLP-1 receptor agonist carries a boxed warning about thyroid C-cell tumors based on rodent studies, in which sustained GLP-1 receptor activation produced medullary thyroid carcinomas in rats and mice. [3][7] Whether this translates to humans is unresolved. Large observational analyses have produced mixed signals, and no human causal link has been established. The FDA has nonetheless made the contraindication explicit:
- Personal or family history of medullary thyroid carcinoma (MTC)
- Multiple Endocrine Neoplasia syndrome type 2 (MEN2)
For patients without those histories, the consensus among endocrinology societies is that the absolute risk is very low. Routine calcitonin or thyroid ultrasound screening is not currently recommended. Symptoms that should prompt an urgent thyroid evaluation include a new neck mass, persistent hoarseness, or trouble swallowing, although these complaints are far more often caused by something benign. [3]
Bottom line: if you have MTC or MEN2 in your family, GLP-1s are off the table. Otherwise the rodent finding informs the warning but is not a reason most patients should avoid the class.
Hypoglycemia, kidney injury, and retinopathy
Used alone, GLP-1 agonists rarely cause hypoglycemia because they stimulate insulin only when blood glucose is elevated. The risk rises sharply when they are combined with insulin or sulfonylureas, in which case dose reduction of the partner drug is standard. [3] Patients on metformin alone or no other glucose-lowering medication essentially do not see hypoglycemia from a GLP-1.
Acute kidney injury appears as a precaution because severe nausea, vomiting, or diarrhea can dehydrate patients enough to compromise kidney perfusion, occasionally requiring hospitalization or temporary dialysis. A FAERS pharmacovigilance analysis identified 2,670 cases of GLP-1-associated AKI, with liraglutide accounting for 35% of reports, a median time-to-onset of 63 days, and a hospitalization rate near 45%. [17] The fix is supportive: hydrate aggressively, hold the drug if intake cannot keep up with losses, and call a clinician if urine output drops or dizziness on standing persists. Patients with chronic kidney disease, on diuretics, or on ACE inhibitors / ARBs should be especially careful during titration.
Diabetic retinopathy complications are a specific warning for patients with type 2 diabetes. In the SUSTAIN-6 cardiovascular outcomes trial, semaglutide was associated with a higher rate of diabetic retinopathy complications (3.0% vs 1.8% on placebo), thought to reflect the well-known phenomenon of acute worsening when chronic hyperglycemia is corrected quickly. [3][18] Patients with pre-existing retinopathy should have an eye exam before starting and ongoing ophthalmologic monitoring through the first year.
A separate visual concern is non-arteritic anterior ischemic optic neuropathy (NAION). A 2024 Mass Eye and Ear cohort study published in JAMA Ophthalmology found semaglutide use associated with an increased risk of NAION in patients with diabetes (hazard ratio in the 4-fold range), although follow-up multinational analyses produced inconsistent signals. [19] The European Medicines Agency reviewed the data in 2025 and concluded NAION is a very rare side effect of semaglutide, potentially affecting up to 1 in 10,000 users, and asked manufacturers to add it to product information. [20] Sudden painless vision loss in one eye warrants immediate ophthalmology evaluation.
Bottom line: these risks are concentrated in specific subgroups (insulin/sulfonylurea users, patients with retinopathy, anyone unable to keep down fluids). Routine GLP-1 use without those factors carries low risk for any of them.
Mood, suicidal ideation, and depression
Postmarketing reports of suicidal ideation triggered an FDA review beginning in 2023. In its January 2024 update, the FDA stated that a preliminary evaluation had not found evidence that GLP-1 use causes suicidal thoughts or actions, while noting the review would continue. [21] An NIH-funded analysis published the same month found that semaglutide users had a 49% to 73% lower risk of new suicidal ideation than patients on other obesity or diabetes treatments, and a 2024 European Medicines Agency review reached the same negative-signal conclusion. [22]
In January 2026, after a meta-analysis of 91 trials covering more than 107,000 participants and a retrospective study of 2.2 million adults with diabetes both found no increased risk, the FDA requested removal of the suicidal ideation warning from labels for liraglutide (Saxenda), semaglutide 2.4 mg (Wegovy), and tirzepatide (Zepbound). [23] Patients with active depression or suicidal ideation should still be monitored, both because depression itself is common in obesity and because rapid life changes can destabilize mental health, but current evidence does not support a causal drug effect. Patients should report any new or worsening mood symptoms to their prescriber regardless of the warning status.
Pregnancy, breastfeeding, and contraception
GLP-1 agonists are contraindicated in pregnancy. Animal data show fetal harm at maternally toxic doses, and human data are insufficient to rule out risk. Manufacturers and most clinicians recommend a washout period before conception of at least 2 months for semaglutide and tirzepatide given their long half-lives (about 1 week each, so roughly 5 to 6 half-lives clear the drug). [3][7][24] Liraglutide, with a half-life of about 13 hours, clears within days. Effective contraception during treatment is standard guidance.
Tirzepatide carries an additional contraception caveat: because it slows gastric emptying more aggressively than pure GLP-1 drugs, it can reduce the absorption and effectiveness of oral contraceptives. A pharmacokinetic study showed peak concentrations of ethinyl estradiol and norgestimate dropping by roughly 55% to 66% after a single 5 mg tirzepatide dose. The Zepbound label recommends switching to a non-oral contraceptive method or adding a barrier method for 4 weeks after starting tirzepatide and for 4 weeks after each dose increase. [25] Other GLP-1s have not shown this contraceptive interaction.
Breastfeeding data are limited. Manufacturers generally recommend discontinuation while nursing, although peptide drugs are unlikely to be absorbed orally by an infant. Most clinicians counsel weighing the maternal benefit against limited safety data and individualizing the decision.
Bottom line: the suicidal ideation signal has not held up under formal review. Pregnancy is a hard contraindication and requires planning ahead, especially on tirzepatide where oral contraceptive failure adds an extra layer.
Special populations: adolescents, older adults, and eating disorders
Wegovy is FDA-approved for adolescents aged 12 and older with obesity, and Saxenda from age 12 with obesity. The STEP TEENS trial of semaglutide 2.4 mg in 201 adolescents reported the same dominant GI side-effect pattern as the adult trials, with nausea, vomiting, or diarrhea in 62% of the semaglutide arm versus 42% of placebo. Cholelithiasis was reported in 3.8% of treated adolescents (versus 0% on placebo), a higher rate than in adult trials. Serious adverse events occurred in 11% of the semaglutide group versus 9% of placebo. Long-term safety data in growing patients remain limited, and pediatric prescribing is typically reserved for specialty obesity clinics. [26]
Older adults metabolize the drugs normally, but they are more vulnerable to dehydration-related kidney injury, pre-existing gastroparesis, and sarcopenia. Slower titration, careful protein and fluid intake, and resistance training matter even more after 65.
Patients with active or historic eating disorders deserve careful evaluation before starting. The drug's appetite suppression can entrench restrictive patterns or mask binge-purge cycles, and the rapid weight change can destabilize body-image disorders. Eating-disorder specialty centers are increasingly treating "GLP-1-induced anorexia nervosa" as a pattern, and most obesity-medicine guidelines advise screening for eating disorders before prescribing.
Allergic reactions and rare events
Hypersensitivity reactions including rash, urticaria, angioedema, and rarely anaphylaxis are reported across the class. With tirzepatide, hypersensitivity occurred in 5.5% of patients who developed anti-tirzepatide antibodies versus 3.0% who did not, with most reactions being mild skin findings. [4] Anaphylaxis is rare but documented; immediate medical attention is appropriate for facial swelling, throat tightness, or trouble breathing after a dose.
Other rare-but-warning-level events documented in postmarketing reports include acute angle-closure glaucoma, transient elevations in liver enzymes, and isolated case reports of pancreatic cancer that have not held up in case-control analyses. [3][6] These remain rare enough that screening is not recommended in the absence of symptoms.
What happens when you stop: weight regain and rebound
GLP-1s suppress appetite through pharmacology. When the drug clears, the appetite signal returns. The STEP-1 extension followed patients after a 68-week treatment phase ended and showed that participants regained roughly two-thirds of the weight they had lost within one year of stopping semaglutide. By week 120 (52 weeks off drug), the semaglutide arm had regained a mean of 11.6 percentage points of weight loss versus 1.9 in the placebo arm, leaving net losses from baseline of 5.6% versus 0.1%. [27] Cardiometabolic markers (HbA1c, lipids, CRP) regressed only partially, retaining some benefit despite weight regain.
For type 2 diabetes, glucose control typically deteriorates within weeks to months of stopping unless other agents are added. For weight loss alone, the regain trajectory in real-world data appears slower than in trial extensions, possibly because patients in practice taper, switch doses, or adopt complementary lifestyle changes rather than stopping cleanly. Either way, the practical model is closer to "treatment for a chronic condition" than "course of medication," similar to antihypertensives. Plans to taper off should include a structured maintenance plan: continued protein and resistance training, a dietary plan that does not rely on the drug for portion control, and follow-up weight monitoring with a clear threshold for reinitiating treatment.
Managing side effects: practical steps
Most GLP-1 side effects are dose-related and time-limited. The standard playbook for getting through them:
- Titrate slowly. Starting low and stretching out dose increases reduces GI events more reliably than any anti-nausea medication.
- Eat smaller, more frequent meals. Stop when you feel about 70% full.
- Skip greasy, fried, very rich, or strongly spiced food on injection day and the day after.
- Hydrate. Aim for at least 2 liters of water daily; more if vomiting or diarrhea is occurring.
- Prioritize protein (1.2 to 1.6 g/kg/day) and resistance training to protect lean mass.
- Keep a stool softener and an antacid available; use them early rather than waiting.
- For nausea that does not respond to diet changes, ondansetron 4 mg (prescription) on demand is the most commonly used antiemetic, with metoclopramide reserved because of its own GI motility effects.
- Hold the drug, do not push through, if you cannot keep fluids down for more than 24 hours.
- If you missed a weekly dose by more than 5 days, restart at the next scheduled dose rather than doubling up.
Call a clinician promptly for: severe abdominal pain that radiates to the back, persistent vomiting, signs of dehydration (low urine output, dizziness on standing), neck swelling or hoarseness, jaundice, sudden vision changes in one eye, signs of allergic reaction (face swelling, hives, breathing trouble), or any symptom that feels out of proportion to "normal" GI upset.
Bottom line: the difference between a tolerable GLP-1 experience and a miserable one is usually titration speed, eating habits, and hydration, not the specific drug.
How long do side effects last and when to expect them
Timing is one of the most useful pieces of information for patients deciding whether to push through a side effect or call the prescriber. Across STEP, SUSTAIN, SURMOUNT, and PIONEER trials, the same temporal pattern repeats: nausea peaks in the first 1 to 2 weeks at each new dose, declines over the following 4 to 6 weeks, and largely disappears by week 8 to 12 once the dose is held steady. Vomiting follows a similar curve but with lower absolute incidence. Diarrhea and constipation are more variable: some patients alternate between the two as the gut adapts, and bowel pattern often does not stabilize until week 12 or beyond. [1][2]
Less common timing patterns to know:
- Hair shedding typically starts 8 to 16 weeks after rapid weight loss begins, peaks around month 4 to 6, and recovers spontaneously by month 9 to 12.
- Gallstones cluster between months 2 and 12, when weight-loss velocity is highest.
- Pancreatitis events have no characteristic onset window; they have been reported from the first dose to several years into therapy.
- Gastroparesis-style fullness usually announces itself within the first 1 to 3 months and worsens with dose escalation; if persistent fullness appears late in treatment, look for another cause first.
- Acute kidney injury from dehydration usually follows a discrete vomiting or diarrhea episode by 1 to 5 days, with median time-to-event around 9 weeks into therapy in FAERS data. [17]
- Retinopathy worsening in T2D often appears in the first 6 months after rapid HbA1c improvement, then stabilizes.
The takeaway: a symptom that appeared with a dose increase and is improving week-over-week is usually safe to ride out with supportive care. A symptom that started long after the last dose change, or is worsening despite dose stability, deserves a fresh look from the prescriber.
Side effects vs intended effects: where the line blurs
Some "side effects" are really the intended pharmacology showing up in places patients did not expect. Decreased appetite, early satiety, and reduced food noise are the desired clinical effects of GLP-1 receptor activation; they appear as adverse events on labels because trial protocols capture every patient-reported change. The same is true of mild taste changes (favorite foods losing appeal), reduced alcohol craving, and the often-positive blunting of impulsive snacking. Patients sometimes interpret these as side effects to call about; clinicians usually do not adjust the dose for them.
The flip side is that the intended pharmacology can produce unintended quality-of-life effects. Reduced food enjoyment matters socially, especially around meals and celebrations. The "I'm not hungry but I should eat" experience can lead to under-eating and protein deficits if not actively managed. And for some patients with prior disordered eating, the appetite suppression can revive restrictive patterns that needed years of recovery work to manage. None of these are dangerous in the strict pharmacological sense, but they are reasons to talk to a prescriber rather than push through silently.
Frequently asked questions
Are GLP-1 side effects permanent? Almost never. The common GI effects fade within weeks at a stable dose. Hair loss and skin laxity follow the weight-loss curve and recover or stabilize once weight is steady. Pancreatitis, gallbladder disease, and severe gastroparesis can have lasting consequences but are uncommon.
Can I drink alcohol on a GLP-1? No formal contraindication, but many patients lose tolerance and find alcohol amplifies nausea. Hypoglycemia risk is mild on the drug alone, more significant if combined with insulin or sulfonylureas. Some users report markedly reduced cravings, which is being studied formally.
Does taking the drug at night reduce nausea? No high-quality data, but anecdotal reports suggest evening dosing helps some people sleep through the worst of it. Switch the day or time of week if symptoms cluster predictably.
Will I have to stay on it forever? If used for obesity, current evidence supports treating obesity as a chronic disease, with appetite returning when the drug stops. Tapering, switching to a maintenance dose, or pairing with intensive lifestyle support are all being studied. Decisions should be made with a clinician.
Should I get a baseline thyroid ultrasound or calcitonin? Not routinely. Screening is not recommended in patients without MTC or MEN2 history.
Is "Ozempic face" reversible? The volume loss reflects body composition. Some patients regain fullness with modest weight regain or fat-stabilizing strategies. Cosmetic interventions (filler, fat transfer) are an option but should be discussed when weight is stable.
References
- Wilding JPH et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP-1). New England Journal of Medicine. 2021;384:989-1002. doi:10.1056/NEJMoa2032183.
- Jastreboff AM et al. Tirzepatide Once Weekly for the Treatment of Obesity (SURMOUNT-1). New England Journal of Medicine. 2022;387:205-216. doi:10.1056/NEJMoa2206038.
- U.S. Food and Drug Administration. Ozempic (semaglutide) Prescribing Information. Novo Nordisk. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/209637s025lbl.pdf
- U.S. Food and Drug Administration. Zepbound (tirzepatide) Prescribing Information. Eli Lilly. https://www.accessdata.fda.gov/drugsatfda_docs/label/2025/217806s031lbl.pdf
- U.S. Food and Drug Administration. Rybelsus (semaglutide) tablets Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2026/213051s030lbl.pdf
- Marso SP et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes (LEADER). New England Journal of Medicine. 2016;375:311-322. doi:10.1056/NEJMoa1603827.
- U.S. Food and Drug Administration. Wegovy (semaglutide) Prescribing Information. Novo Nordisk. https://www.accessdata.fda.gov/drugsatfda_docs/label/2024/215256s011lbl.pdf
- Cleveland Clinic / dermatology literature. Hair loss and GLP-1 receptor agonists: telogen effluvium pattern. Systematic review of GLP-1 therapies and hair loss. https://pmc.ncbi.nlm.nih.gov/articles/PMC13100445/
- UCLA Health. Ozempic face (and other GLP-1 side effects). https://www.uclahealth.org/news/article/ozempic-face-and-other-glp-1-side-effects
- Wilding JPH et al. Impact of Semaglutide on Body Composition in Adults with Overweight or Obesity: Exploratory Analysis of the STEP 1 Study. Obesity (Silver Spring). 2021. https://pmc.ncbi.nlm.nih.gov/articles/PMC8089287/
- Look M et al. Body composition changes during weight reduction with tirzepatide in the SURMOUNT-1 study. Diabetes, Obesity and Metabolism. 2025. doi:10.1111/dom.16275.
- Sodhi M, Rezaeianzadeh R, Kezouh A, Etminan M. Risk of Gastrointestinal Adverse Events Associated With Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss. JAMA. 2023;330(18):1795-1797. doi:10.1001/jama.2023.19574.
- He L et al. Association of GLP-1 Receptor Agonist Use With Risk of Gallbladder and Biliary Diseases: A Systematic Review and Meta-analysis. JAMA Internal Medicine. 2022;182(5):513-519. doi:10.1001/jamainternmed.2022.0338.
- U.S. Judicial Panel on Multidistrict Litigation. In re: Glucagon-Like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation, MDL No. 3094. https://www.jpml.uscourts.gov/
- American Society of Anesthesiologists. Consensus-based guidance on preoperative management of patients on glucagon-like peptide-1 receptor agonists. June 2023. https://www.asahq.org/about-asa/newsroom/news-releases/2023/06/american-society-of-anesthesiologists-consensus-based-guidance-on-preoperative
- Multisociety Clinical Practice Guidance for the Safe Use of Glucagon-like Peptide-1 Receptor Agonists in the Perioperative Period. October 2024. Clinical Gastroenterology and Hepatology. https://www.cghjournal.org/article/S1542-3565(24)00910-8/fulltext
- Zhou et al. Glucagon-like peptide-1 receptor agonists and acute kidney injury: a FAERS pharmacovigilance analysis. Frontiers in Endocrinology. 2022. https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2022.1032199/full
- Marso SP et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (SUSTAIN-6). New England Journal of Medicine. 2016;375:1834-1844. doi:10.1056/NEJMoa1607141.
- Hathaway JT et al. Risk of Nonarteritic Anterior Ischemic Optic Neuropathy in Patients Prescribed Semaglutide. JAMA Ophthalmology. 2024. https://jamanetwork.com/journals/jamaophthalmology/fullarticle/2820255
- European Medicines Agency / WHO. The use of semaglutide medicines and risk of NAION. June 2025. https://www.who.int/news/item/27-06-2025-27-06-2025-semaglutide-medicines-naion
- U.S. Food and Drug Administration. Update on FDA's ongoing evaluation of reports of suicidal thoughts or actions in patients taking certain type of medicines approved for type 2 diabetes and weight loss. January 2024. https://www.fda.gov/drugs/drug-safety-and-availability/update-fdas-ongoing-evaluation-reports-suicidal-thoughts-or-actions-patients-taking-certain-type
- Wang W et al. Association of semaglutide with risk of suicidal ideation in a real-world cohort. Nature Medicine. 2024. https://www.nature.com/articles/s41591-023-02672-2
- U.S. Food and Drug Administration. FDA Requests Removal of Suicidal Behavior and Ideation Warning from GLP-1 Receptor Agonist (GLP-1 RA) Labeling. January 2026. https://www.fda.gov/drugs/drug-safety-communications/fda-requests-removal-suicidal-behavior-and-ideation-warning-glucagon-peptide-1-receptor-agonist-glp
- MotherToBaby. Semaglutide fact sheet. National Library of Medicine Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK600385/
- Jensen SBK et al. The impact of tirzepatide and glucagon-like peptide 1 receptor agonists on oral hormonal contraception. Journal of the American Pharmacists Association. 2023. https://www.japha.org/article/S1544-3191(23)00370-9/fulltext
- Weghuber D et al. Once-Weekly Semaglutide in Adolescents with Obesity (STEP TEENS). New England Journal of Medicine. 2022;387:2245-2257. doi:10.1056/NEJMoa2208601.
- Wilding JPH et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes, Obesity and Metabolism. 2022. doi:10.1111/dom.14725.