Retatrutide: Triple Agonist for Weight Loss
Summary: Retatrutide (Eli Lilly code LY3437943) is an investigational once-weekly injectable that activates three gut hormone receptors at once: GIP, GLP-1, and glucagon. It is not FDA-approved and is not commercially available; the first phase 3 readout (TRIUMPH-4) landed in December 2025, with the rest of the registrational program expected to report through 2026.
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.
What retatrutide is
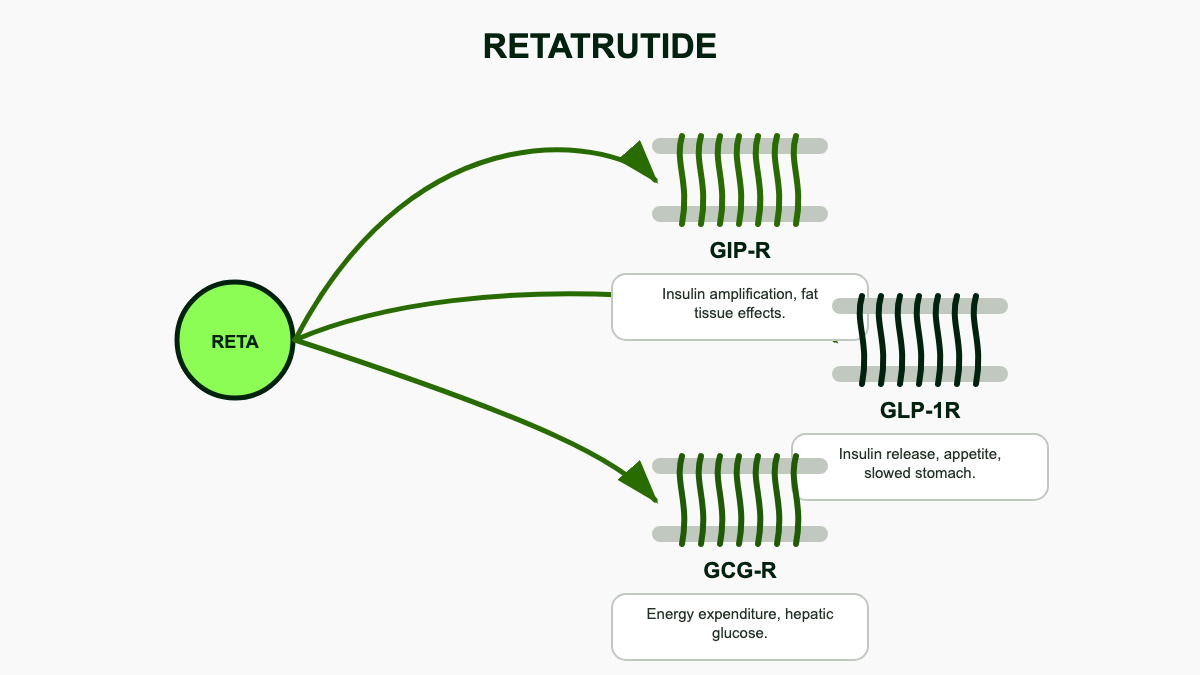
Retatrutide is Eli Lilly's experimental obesity and metabolic disease drug, internally known as LY3437943. It is a single peptide engineered to bind and activate three receptors at once: the glucose-dependent insulinotropic polypeptide (GIP) receptor, the glucagon-like peptide-1 (GLP-1) receptor, and the glucagon receptor. [1][5] Semaglutide hits one of those targets (GLP-1). Tirzepatide hits two (GIP and GLP-1). Retatrutide adds the glucagon receptor as a third lever, which is why it is often called a "triple agonist" or "triple G."
The drug is given as a once-weekly subcutaneous injection, with doses titrated upward over several months to limit gastrointestinal side effects. As of May 2026 it is still investigational; no version of retatrutide is approved by the FDA, EMA, or any other major regulator, and there is no legal pharmacy supply outside of Lilly-sponsored clinical trials. [5]
Structurally, retatrutide is a 39-amino-acid peptide built on a modified GIP backbone with a fatty diacid side chain that prolongs half-life and supports once-weekly dosing. Its receptor activity is intentionally lopsided. The molecule is most potent at the GIP receptor, less potent at GLP-1, and lower still at glucagon, with the glucagon component dialed in deliberately to add metabolic effect without causing the persistent hyperglycemia that pure glucagon agonists produce. [1][5][7] That balance is the central design choice behind the drug.
Why adding the glucagon receptor matters
Glucagon is usually thought of as the hormone that raises blood sugar, which sounds like the opposite of what you want in a diabetes or obesity drug. The full picture is more interesting. Sustained, low-level glucagon receptor activation appears to increase resting energy expenditure and push the liver to burn fat, which translates into more weight loss per unit of appetite suppression than GLP-1 alone can deliver. [1][5] It is the same logic behind survodutide, Boehringer Ingelheim's dual GLP-1/glucagon agonist, but retatrutide layers GIP on top.
The hepatic effects matter beyond weight. Glucagon receptor activation drives lipolysis and beta-oxidation in the liver, which is why retatrutide produces unusually large reductions in liver fat at modest doses. In a phase 2a substudy of adults with metabolic dysfunction-associated steatotic liver disease (MASLD), 48 weeks of retatrutide 12 mg cut liver fat by about 86%, and roughly 93% of participants on that dose reached a normal liver-fat threshold below 5%. [8] The 8 mg dose produced an 81.7% relative reduction with around 89% of participants normalizing. Those numbers are striking because liver fat reduction in MASLD trials of pure GLP-1 drugs typically tops out in the 30% to 50% range over similar follow-up.
The same mechanism explains why retatrutide nudges resting metabolic rate upward and why it produces meaningful changes in lipids and inflammation markers that go beyond what is expected from weight loss alone. The trade-off is that glucagon agonism can theoretically blunt glycemic improvement, which is why early dose-finding work in type 2 diabetes paid such close attention to HbA1c. The published data so far show the GIP and GLP-1 components more than offset that risk in real participants.
Phase 2 results: the 24% number
The defining phase 2 readout for retatrutide is the trial published by Jastreboff and colleagues in the New England Journal of Medicine in 2023. [1] It enrolled 338 adults with a BMI of at least 30, or 27 with weight-related conditions, and randomized them to placebo or one of several retatrutide doses (1 mg, 4 mg, 8 mg, or 12 mg weekly) for 48 weeks.
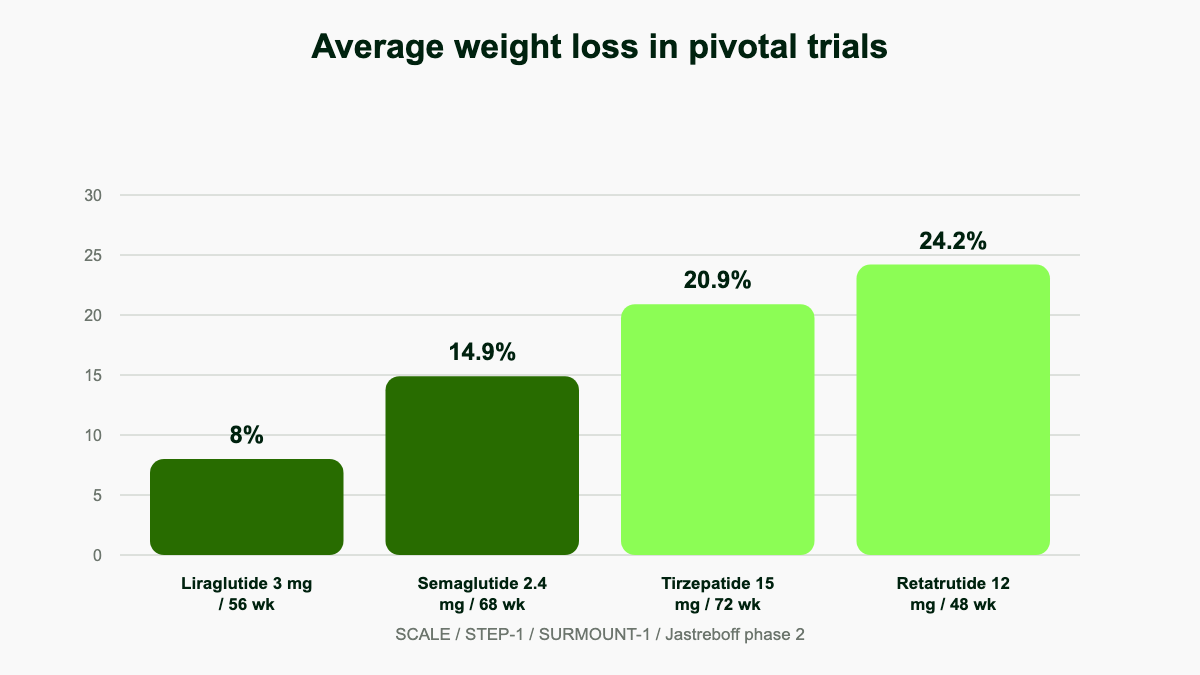
Mean weight change at 48 weeks:
- Placebo: -2.1%
- 1 mg: -8.7%
- 4 mg: -17.1%
- 8 mg: -22.8%
- 12 mg: -24.2%
For context, the STEP 1 trial of semaglutide 2.4 mg produced about 14.9% weight loss at 68 weeks, and the SURMOUNT-1 trial of tirzepatide 15 mg reached roughly 20.9% at 72 weeks. Retatrutide's 24.2% came in 48 weeks, and the curves had not yet plateaued. [1]
At the 4 mg dose, 92% of participants lost at least 5% of body weight, 75% lost at least 10%, and 60% lost at least 15%. [1] At 24 weeks the corresponding figures were already -7.2% in the 1 mg arm, -12.9% at 4 mg, -17.3% at 8 mg, and -17.5% at 12 mg, ahead of where most GLP-1 monotherapies sit at the same time point. [1] Side effects were largely what the GLP-1 class has trained clinicians to expect: nausea, diarrhea, vomiting, and constipation, mostly mild to moderate, dose-related, and somewhat blunted by starting at 2 mg instead of 4 mg. Heart rate rose modestly during titration and then drifted back down. No unexpected safety signal emerged in phase 2.
Phase 2 in type 2 diabetes
The companion phase 2 trial in type 2 diabetes, published in The Lancet alongside the obesity readout, gave a clearer view of the drug's metabolic profile. It randomized 281 adults with poorly controlled type 2 diabetes (mean baseline HbA1c around 8.3%) to placebo, dulaglutide 1.5 mg, or one of several retatrutide doses for 36 weeks. [9]
HbA1c reductions at 24 weeks landed at -0.43% on retatrutide 0.5 mg, -1.39% to -1.30% across the 4 mg arms, -1.99% to -1.88% across the 8 mg arms, and -2.02% on retatrutide 12 mg, against -1.41% for dulaglutide and -0.01% for placebo. [9] The 8 mg and 12 mg doses outperformed dulaglutide. Up to 82% of retatrutide participants pushed HbA1c below 6.5%, and roughly 31% reached 5.7%, the threshold for normoglycemia. Body weight fell by about 16.94% on the 12 mg dose at 36 weeks, against 2.02% on dulaglutide and 3.0% on placebo. [9] That dataset settled the early concern that adding a glucagon agonist might compromise glycemic control. It did not.
The diabetes trial also produced one of the cleaner views of the drug's effect on lipids and inflammation. By 24 weeks, retatrutide doses at 4 mg and above lowered triglycerides by 35.4% to 40.0%, leptin by 29.0% to 55.8%, and FGF-21 by 52.2% to 65.7%, while raising beta-hydroxybutyrate, a circulating marker of fatty-acid oxidation. [8] That biochemical fingerprint is consistent with glucagon-driven hepatic lipolysis on top of GLP-1-driven appetite suppression, which is exactly what the molecule was designed to do. The same trial reported nausea in roughly a third of participants on the higher target doses, similar to the obesity trial, with vomiting and diarrhea following the same dose-dependent pattern.
The TRIUMPH phase 3 program
Lilly's registrational program for retatrutide is called TRIUMPH. It is built around four pivotal weight-management trials and several supporting indication studies that read out on a staggered schedule through 2026 and 2027. The four lead studies are:
- TRIUMPH-1 (NCT05929066): roughly 2,300 adults with obesity or overweight, evaluating retatrutide 4 mg, 9 mg, and 12 mg over 80 weeks. The trial includes nested substudies in knee osteoarthritis and obstructive sleep apnea so a single registrational program can support multiple label claims. Primary endpoint is percent change in body weight at week 80. Readout is targeted for the second or third quarter of 2026. [4][10]
- TRIUMPH-2 (NCT05929079): retatrutide in adults with obesity or overweight and type 2 diabetes, again at 4 mg, 9 mg, and 12 mg, with an OSA substudy nested inside. Phase 2 produced HbA1c reductions of up to 2.0% in this population, so TRIUMPH-2 is the trial that turns those numbers into a regulatory dataset. Readout is also expected in 2026. [10][11]
- TRIUMPH-3 (NCT05931367): a weight-management trial in adults with obesity and established cardiovascular disease, testing 9 mg and 12 mg. It is not the dedicated cardiovascular outcomes trial, but it is the first phase 3 dataset in patients with prior CV events and will inform whether retatrutide is safe to use in that population pending the larger outcomes study. [6]
- TRIUMPH-4 (NCT05931380): 445 adults with obesity and knee osteoarthritis, randomized 1:1:1 to placebo, retatrutide 9 mg, or 12 mg for 68 weeks. This is the trial that has already reported. [2]
Beyond the four headline trials, Lilly is running additional phase 3 work that will broaden the eventual label. TRIUMPH-Outcomes (NCT06383390) is the cardiovascular and kidney outcomes trial in adults with obesity plus established atherosclerotic CV disease or chronic kidney disease, and is event-driven rather than time-driven; readout is widely expected in 2027 or 2028. [12] TRANSCEND-CKD (NCT05936151) is a phase 2b mechanistic study using iohexol-clearance measured GFR to look at how retatrutide changes kidney function in adults with overweight or obesity and CKD with or without type 2 diabetes. [13] A separate phase 3 trial is studying retatrutide in MASH/MASLD. Two additional studies cover moderate-to-severe obstructive sleep apnea and chronic low back pain. [10] Together the program enrolled more than 5,800 participants across the four registrational weight-management studies alone, with seven additional phase 3 readouts expected through 2026. [2]
TRIUMPH-4: the first phase 3 readout
On December 11, 2025, Lilly announced topline results from TRIUMPH-4, the first phase 3 trial of retatrutide to report. [2] Participants entered with an average weight of 112.7 kg (about 248.5 lb) and a mean BMI of 40.4 kg/m², and 84% started with a BMI of 35 or higher. After 68 weeks of treatment in adults with obesity and knee osteoarthritis:
- Retatrutide 12 mg: 28.7% mean weight loss (about 32.3 kg, 71.2 lb)
- Retatrutide 9 mg: 26.4% mean weight loss (about 29.1 kg, 64.2 lb)
- Placebo: 2.1% mean weight loss (about 4.6 lb)
Both co-primary endpoints were met. WOMAC knee pain scores fell by 4.5 points on retatrutide 9 mg (a 75.8% improvement from baseline) and 4.4 points on 12 mg (74.3%), against 2.4 points on placebo. [2][3] Roughly 14.1% of participants on 9 mg and 12.0% on 12 mg reported complete pain resolution at week 68, against 4.2% on placebo. [2] WOMAC physical function improved by 71.8% to 73.7% on retatrutide. The depth of weight loss in TRIUMPH-4 was unusual: 47.7% of the 9 mg arm and 58.6% of the 12 mg arm hit at least 25% loss, 30.5% and 39.4% reached 30%, and 18.2% and 23.7% crossed 35%. [2] Less than 1% of placebo participants hit 25%.
The cardiometabolic readouts were equally striking for an obesity drug. The 12 mg dose lowered systolic blood pressure by 14.0 mmHg from baseline, alongside reductions in non-HDL cholesterol, triglycerides, and high-sensitivity C-reactive protein. [2] The most common side effects were the familiar GI cluster: nausea (38.1% on 9 mg, 43.2% on 12 mg, 10.7% on placebo), diarrhea (34.7%, 33.1%, 13.4%), constipation (21.8%, 25.0%, 8.7%), and vomiting (20.4%, 20.9%, 0%). [3] Discontinuations from adverse events ran 12.2% at 9 mg and 18.2% at 12 mg, against 4.0% on placebo, with some reportedly tied to perceived excessive weight loss. [3][14]
One side effect did not appear in phase 2: dysesthesia, an abnormal sensation of touch or burning, was reported in 8.8% of the 9 mg arm and 20.9% of the 12 mg arm, against 0.7% on placebo. [14] It is not severe enough to derail the program, but it is a new entry on retatrutide's safety ledger and one analysts will be tracking through the rest of the TRIUMPH readouts. TRIUMPH-4 is one trial in one population, but it confirmed the phase 2 magnitude in a much larger, longer, properly controlled study. The 28.7% figure also became the first phase 3 result in the GLP-1 class to clear SURMOUNT-1's tirzepatide numbers in a broadly comparable design.
Retatrutide vs tirzepatide vs semaglutide
Direct head-to-head trials between retatrutide and tirzepatide or semaglutide have not yet reported. The cross-trial comparison is rough, but consistent:
- Semaglutide 2.4 mg (Wegovy): about 14.9% weight loss at 68 weeks in STEP 1.
- Tirzepatide 15 mg (Zepbound): about 20.9% at 72 weeks in SURMOUNT-1.
- Retatrutide 12 mg: 24.2% at 48 weeks in phase 2; 28.7% at 68 weeks in TRIUMPH-4. [1][2]
A 2025 network meta-analysis that pooled the available randomized data put retatrutide at roughly -23.8% mean change vs around -16.8% for tirzepatide across comparable indirect comparisons, mirroring the headline trial figures. [15] The trade-off is gastrointestinal tolerability and discontinuation rates, which are higher at the top retatrutide doses than at top tirzepatide doses in their respective trials. Whether the 9 mg dose can deliver most of the weight loss with fewer dropouts is a question the rest of the TRIUMPH program will help answer. The 9 mg arm in TRIUMPH-4 lost 26.4% with a 12.2% discontinuation rate, putting it in roughly the same ballpark as tirzepatide's tolerability profile while still beating its peak efficacy.
The other practical difference is mechanism risk. Tirzepatide and semaglutide are well-studied molecules with multiple completed cardiovascular outcomes trials between them. Retatrutide is the first triple agonist to reach pivotal-stage data, and its glucagon-receptor component has no long-term outcomes record yet. Until TRIUMPH-Outcomes reads out, comparisons should treat retatrutide as the drug with the better short-term efficacy and the larger long-term unknown.
Dosing, pharmacology, and what to expect on therapy
Retatrutide is a once-weekly subcutaneous injection. Across the phase 3 program Lilly has used a slow titration schedule: 2 mg starting dose, with stepwise increases every four weeks until the assigned target dose. TRIUMPH-4 tested 9 mg and 12 mg; TRIUMPH-1 and TRIUMPH-2 add a 4 mg maintenance arm to test whether a lower dose retains most of the weight loss with better tolerability. [2][10] The eventual commercial dose ladder has not been finalized, but the most likely framing is a stepwise titration from 2 mg up to 4 mg, 9 mg, or 12 mg, with the target chosen based on tolerability and clinical response.
The pharmacokinetics support weekly dosing comfortably. Retatrutide's terminal half-life is roughly six days, similar to tirzepatide, and steady state is reached after about four to five weeks at a fixed dose. The fatty diacid side chain on the peptide drives reversible binding to serum albumin, which is what slows clearance enough to make once-weekly injection clinically reasonable. The titration interval of four weeks is therefore set by tolerability, not pharmacokinetics: each step is held long enough to let GI adverse events settle before the next dose escalation.
Average heart rate increases of about 5 to 10 beats per minute are typical at higher doses during titration, peaking around week 24 before drifting back toward baseline. [1] In the phase 2 program, supraventricular arrhythmias were reported in 4% to 14% of retatrutide arms compared with 2% to 3% on placebo, mostly mild and not classified as serious. [1] One participant on the 12 mg arm experienced a serious adverse event of prolonged QT in the context of concurrent ondansetron use, which is the kind of interaction prescribers will want to keep an eye on at the higher doses. Resting metabolic rate appears to rise on retatrutide more than would be predicted from weight loss alone, which is the practical signature of glucagon-receptor engagement.
Body composition data from a phase 2 substudy in type 2 diabetes showed that the loss is preferentially fat: by DXA, fat mass dropped substantially more than lean mass at higher doses, and the ratio compared favorably to what has been published with semaglutide. [16] That detail matters because the durability of weight loss after stopping any GLP-1-class drug depends in part on how much of the loss came from muscle, and retatrutide appears to spare lean tissue at least as well as the existing class. In the same substudy, visceral adipose tissue declined disproportionately compared with subcutaneous fat, which is the body-composition signature most associated with cardiometabolic benefit.
Practical patient-facing details from the trial program: injections are administered subcutaneously into the abdomen, thigh, or upper arm; the device used in TRIUMPH is a multi-dose pen for blinding purposes; missed doses can typically be taken within a few days of the scheduled day without restarting titration; and standard antiemetic strategies (small meals, hydration, slow eating, occasional ondansetron) appear to manage the early GI period adequately for most participants. None of this constitutes a finalized prescribing recommendation, since the label has not been written.
Expected approval timeline
Retatrutide is not approved anywhere as of May 2026. Lilly is waiting on the remaining TRIUMPH readouts, particularly TRIUMPH-1 and TRIUMPH-2, before filing with the FDA. With seven additional phase 3 trials expected to report through 2026, [2] most analyst models point to a regulatory submission in late 2026 or the first half of 2027 and an approval decision in mid- to late 2027 under standard review, or earlier under priority review. [17] That timeline roughly mirrors the path tirzepatide took from pivotal data to FDA action in obesity. Cardiovascular outcomes from TRIUMPH-Outcomes will likely arrive after initial approval and shape the eventual label expansion.
EMA and other major regulators typically follow within six to twelve months of an FDA decision in this class. Commercial launch in the United States is most plausibly a late-2027 to first-half-2028 event, again contingent on no negative surprises in the remaining TRIUMPH readouts. [17]
What pricing might look like
Lilly has not announced pricing and any number before approval is speculation. Tirzepatide for obesity (Zepbound) launched in late 2023 at a US list price of around $1,060 per month and currently sits in roughly that range, with payer-negotiated and manufacturer self-pay programs lowering effective cost for many patients. Semaglutide for obesity (Wegovy) entered at a similar list price. There is no obvious reason retatrutide would be priced below tirzepatide given its incremental efficacy advantage, and Lilly will likely position it at parity or modestly above on launch. The bigger variable is payer coverage, which has lagged efficacy data for the entire class and which determines what most patients actually pay. Compounded versions sold by gray-market suppliers are not a legal alternative for retatrutide, as the next section explains.
One pricing wrinkle that did not exist when tirzepatide launched is direct-to-consumer self-pay. Lilly's LillyDirect channel and Novo's NovoCare have set precedent for selling brand obesity drugs in vials at meaningfully lower out-of-pocket prices than autoinjector pens, currently in the $300 to $500 per month range for higher doses. If Lilly carries that model into retatrutide, the effective floor for cash-pay patients without insurance coverage may sit closer to those numbers than to the headline list price. Medicare coverage for obesity indications remains contested at the federal level, which limits the addressable population that can rely on insurance to absorb the list price. None of this is settled, and the eventual launch dynamics will depend on the same payer fights that shape access for the rest of the class.
What is still unknown
The phase 2 and TRIUMPH-4 data are encouraging on efficacy, but several questions remain open:
- Long-term safety beyond 68 weeks. Most published data top out around a year and a half of dosing, and the dysesthesia signal that emerged in TRIUMPH-4 will need to be tracked across the rest of the program. [14]
- Cardiovascular outcomes. TRIUMPH-Outcomes is the trial that will tell us whether the weight loss translates into fewer heart attacks and strokes, the way it eventually did for semaglutide in SELECT and tirzepatide in SURMOUNT-MMO. Readout is most likely 2027 or 2028. [12]
- Kidney outcomes. TRANSCEND-CKD will provide the first mechanistic look at GFR change in CKD patients on retatrutide, and the larger outcomes question rolls into TRIUMPH-Outcomes. [13]
- Durability after stopping. Like other GLP-1-class drugs, weight regain after discontinuation is expected, but the magnitude has not been formally reported for retatrutide.
- Whether the glucagon component has a meaningful hepatic safety signal in longer follow-up, given liver enzyme effects observed with other glucagon agonists. The phase 2 MASLD data look reassuring but are still short. [8]
- Heart rate and arrhythmia. The supraventricular signal in phase 2 was mild but real, and longer follow-up at full target doses is the only way to know whether it stays mild. [1]
A note on availability and "compounded retatrutide"
Because retatrutide is not approved, there is no legal commercial supply through pharmacies in the United States or anywhere else. Vials sold online as "research peptides" or "compounded retatrutide" are not part of any FDA pathway: there is no approved reference listed drug to compound from, no shortage status to invoke, and no quality oversight on what is actually in the bottle.
The FDA has been explicit on this point. In 2025 the agency issued warning letters to six companies, five in the United States and one in Germany, for marketing compounded retatrutide as an unapproved new drug. [18] In March 2026 it followed up with warning letters to more than thirty telehealth companies for misleading marketing of compounded GLP-1 products, retatrutide included. [18] The legal reasoning is straightforward: sections 503A and 503B of the Federal Food, Drug, and Cosmetic Act allow compounding of approved drugs under specific conditions, neither of which applies to a molecule that has never been approved and is not on any shortage list. [18][19]
The risk profile of "research peptide" retatrutide is therefore not the risk profile of the drug being studied in TRIUMPH. Identity, purity, sterility, dose accuracy, and excipient safety are unverified. People interested in retatrutide who meet eligibility criteria can ask their physician about enrollment in an active TRIUMPH-program trial. That is currently the only legitimate way to receive the molecule.
How TRIUMPH-4 changed the framing
The TRIUMPH-4 readout did more than confirm phase 2. It changed what the rest of the program is being judged against. Before December 2025, the working assumption among analysts was 22% to 24% mean weight loss in phase 3, in line with phase 2 and modestly above tirzepatide. The 28.7% number cleared even bullish forecasts, and BMO Capital Markets had previously framed 20% to 23% as the realistic range. [14] That shift creates two separate problems for the rest of the GLP-1 pipeline: first, the tirzepatide ceiling moved; second, the phase 3 readouts for orforglipron, MariTide, and CagriSema now have to be interpreted against the new bar rather than the old one.
It also raises the stakes for TRIUMPH-1 and TRIUMPH-2. TRIUMPH-4 enrolled a population with a high baseline BMI of 40.4 and an even higher proportion of class-3 obesity, both of which tend to amplify weight-loss percentages relative to leaner trial populations. TRIUMPH-1, which targets a broader obesity population without obligatory comorbidities, may produce a slightly lower headline number for purely demographic reasons, even if the underlying drug effect is identical. Whether that lower number is interpreted as disappointment or as expected regression to the mean will depend almost entirely on how Lilly frames the comparison.
The competitive landscape
Retatrutide is the most-watched candidate in the next-generation GLP-1 pipeline, but it is not the only one. The competitive context shapes how much room it has on launch:
- Orforglipron (Lilly, brand name Foundayo): a once-daily oral small-molecule GLP-1 agonist, FDA-approved on April 1, 2026 for adults with obesity or overweight plus a weight-related comorbidity. ATTAIN-1 reported about 11.2% mean weight loss at 72 weeks at the 36 mg dose, less than retatrutide but with the convenience of a pill and no cold-chain requirement. [20]
- CagriSema (Novo Nordisk): a once-weekly fixed-dose combination of semaglutide and the amylin analog cagrilintide. The phase 3 REDEFINE-1 trial reported about 20.4% weight loss; an FDA new drug application was submitted in late 2025, with a likely 2026 decision. [20]
- Survodutide (Boehringer Ingelheim/Zealand): a once-weekly dual GLP-1/glucagon agonist with positive phase 2 data in obesity and MASH and phase 3 trials underway, including a dedicated HFpEF program (EASi-HF). [21]
- Maridebart cafraglutide / MariTide (Amgen): a once-monthly injectable that combines GLP-1 agonism with GIP receptor antagonism, an inverted version of tirzepatide's mechanism. Phase 2 produced 12% to 16% mean weight loss; phase 3 readouts are not expected before 2027. [20]
None of these candidates currently match retatrutide's headline efficacy, but each has its own positioning angle (oral dosing, monthly injection, established CV data via semaglutide, and so on). The market that retatrutide enters will look very different from the one tirzepatide entered in 2023.
Bottom line
Retatrutide produced the largest mean weight loss ever reported in a phase 3 obesity trial, with a tolerability and cardiometabolic profile consistent with the rest of the GLP-1 class plus a modest new safety signal (dysesthesia) and a still-incomplete cardiovascular outcomes record. It is not approved and not legally available outside Lilly's clinical trials, and the compounded versions sold online are unapproved and unregulated. The realistic earliest US approval window is mid- to late 2027, with launch in 2028 if the rest of the TRIUMPH readouts hold the line.
References
- Jastreboff AM, Kaplan LM, Frias JP, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity. N Engl J Med. 2023;389(6):514-526. doi:10.1056/NEJMoa2301972. nejm.org
- Eli Lilly and Company. Lilly's triple agonist, retatrutide, delivered weight loss of up to an average of 71.2 lbs along with substantial relief from osteoarthritis pain in first successful Phase 3 trial. Press release, December 11, 2025. investor.lilly.com
- Rheumatology Advisor. TRIUMPH-4 Results: Retatrutide Cuts Weight and Knee OA Pain. December 2025. rheumatologyadvisor.com
- ClinicalTrials.gov. A Study of Retatrutide (LY3437943) in Adult Participants With Obesity or Overweight (TRIUMPH-1). NCT05929066. clinicaltrials.gov
- Eli Lilly and Company. What to know about retatrutide. lilly.com. lilly.com
- ClinicalTrials.gov. A Study of Retatrutide (LY3437943) Once Weekly in Participants With Obesity and Cardiovascular Disease (TRIUMPH-3). NCT05931367. clinicaltrials.gov
- Wikipedia contributors. Retatrutide. en.wikipedia.org/wiki/Retatrutide
- Sanyal AJ, Kaplan LM, Frias JP, et al. Triple hormone receptor agonist retatrutide for metabolic dysfunction-associated steatotic liver disease: a randomized phase 2a trial. Nat Med. 2024;30(7):2037-2048. nature.com
- Rosenstock J, Frias J, Jastreboff AM, et al. Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial conducted in the USA. Lancet. 2023;402(10401):529-544. pubmed.ncbi.nlm.nih.gov
- Giblin JT, et al. Retatrutide for the treatment of obesity, obstructive sleep apnea and knee osteoarthritis: Rationale and design of the TRIUMPH registrational clinical trials. Diabetes Obes Metab. 2026. dom-pubs.onlinelibrary.wiley.com
- ClinicalTrials.gov. A Study of Retatrutide (LY3437943) in Participants With Type 2 Diabetes Mellitus and Obesity or Overweight (TRIUMPH-2). NCT05929079. clinicaltrials.gov
- ClinicalTrials.gov. The Effect of Retatrutide Once Weekly on Cardiovascular Outcomes and Kidney Outcomes in Adults Living With Obesity (TRIUMPH-Outcomes). NCT06383390. clinicaltrials.gov
- ClinicalTrials.gov. A Study of Retatrutide (LY3437943) on Renal Function in Adults With Obesity and Chronic Kidney Disease (TRANSCEND-CKD). NCT05936151. clinicaltrials.gov
- BioSpace. Lilly's Retatrutide Scores Triple Trial Triumph With 26% Weight Loss, But New Safety Signal Emerges. December 2025. biospace.com
- SUN-659 Comparative Efficacy and Safety of Tirzepatide vs Retatrutide in Weight Loss: A Network Meta-Analysis of Clinical Trials. J Endocr Soc. 2024. pmc.ncbi.nlm.nih.gov
- Effects of retatrutide on body composition in people with type 2 diabetes: a substudy of a phase 2 randomised trial. Lancet Diabetes Endocrinol. 2025. pubmed.ncbi.nlm.nih.gov
- Honest Care. Retatrutide FDA Approval Timeline & Status 2025-2027. findhonestcare.com
- Lengea Law. FDA Warns Companies Over Compounded Retatrutide. 2025. lengealaw.com
- U.S. Food and Drug Administration. FDA's Concerns with Unapproved GLP-1 Drugs Used for Weight Loss. fda.gov
- Prime Therapeutics. GLP-1 Pipeline Update: February 2026. primetherapeutics.com
- Boehringer Ingelheim. EASi-HF Phase III trial in patients with HFpEF. boehringer-ingelheim.com