GLP-1 Dosage Guide
Summary: Every approved GLP-1 receptor agonist uses a stepwise titration that starts well below the therapeutic dose and increases at fixed intervals (4 weeks for weekly injections, 1 week for daily liraglutide, 30 days for oral semaglutide) so the gut can adjust before reaching the maintenance level.
This content is for informational purposes only and is not medical advice. Always consult a qualified healthcare provider before starting, changing, or stopping any medication.
Why every GLP-1 starts at a sub-therapeutic dose
GLP-1 agonists slow gastric emptying and act on central appetite circuits in the hypothalamus and brainstem. Both effects produce nausea, vomiting, diarrhea and constipation when the receptor is hit hard from day one. The 0.25 mg starting dose of semaglutide and the 2.5 mg starting dose of tirzepatide are explicitly labeled as "for treatment initiation" and are not effective for glycemic control or weight loss on their own. They exist solely to let the GI tract adapt before the therapeutic dose arrives. [1][2]
The biology behind the ramp is straightforward. GLP-1 receptors in the gut and brain become less sensitive to the slowing-down signal with continuous exposure, a process called tachyphylaxis. By the end of four weeks at 0.25 mg or 2.5 mg, most patients tolerate the next step with substantially less nausea than if the higher dose had been started cold. Skipping the initiation phase, even for patients who insist they "tolerate everything," produces dropout rates that the manufacturers consider unacceptable for a chronic medication. [3]
The practical consequence is that the first month of any GLP-1 should be judged on tolerance, not on the scale or A1C. If side effects are still rough at the end of an escalation interval, prescribers routinely hold the current dose for another 4 weeks before stepping up rather than abandoning treatment. The label language for both Ozempic and Mounjaro permits "consider delaying dose escalation" if GI symptoms persist, and clinicians often extend a step by 8 to 12 weeks rather than discontinue the drug. [3][4]
The interval choice is not arbitrary. Semaglutide has a half-life of about 168 hours (7 days), so steady state on any new dose is reached in roughly 4 to 5 weeks. Tirzepatide has a slightly shorter half-life of about 5 days, which still means 4 weeks is the minimum interval to reach steady state and assess tolerance. Liraglutide clears within roughly 2 to 3 days, which is why its titration is weekly rather than monthly. Oral semaglutide reaches steady state in 4 to 5 weeks, which matches the 30-day step in the Rybelsus label. Crossing an escalation step before steady state simply means the patient experiences the GI burden of the new dose on top of incomplete adaptation to the previous one. [3]
Ozempic dose chart (semaglutide for type 2 diabetes)
Ozempic is dosed once weekly by subcutaneous injection into the abdomen, thigh or upper arm. The label defines a fixed initiation phase, then offers three possible maintenance doses depending on glycemic response. Sites should be rotated with every dose to avoid lipohypertrophy, and the day of the week can be changed as long as the next injection is at least 48 hours after the previous one. [4]
| Phase | Dose | Duration | Purpose |
|---|---|---|---|
| Initiation | 0.25 mg weekly | 4 weeks | Tolerance only, not therapeutic |
| First therapeutic step | 0.5 mg weekly | At least 4 weeks | Minimum effective dose for A1C reduction |
| Optional step-up | 1 mg weekly | At least 4 weeks before next step | If additional A1C control needed |
| Maximum | 2 mg weekly | Maintenance | Highest FDA-approved Ozempic dose |
The 2 mg dose was added to the Ozempic label in 2022 and is the ceiling: there is no 2.4 mg or higher Ozempic strength, despite that number being common in semaglutide compounding. [4][5] Ozempic is supplied in three multi-dose pens and the strength of the pen determines which doses can be selected. [4]
| Pen | Doses available per click | Number of weekly doses per pen |
|---|---|---|
| 0.25 / 0.5 mg pen (red label) | 0.25 mg or 0.5 mg | 4 doses at 0.25 mg, or 2 doses at 0.5 mg |
| 1 mg pen (blue label) | 1 mg only | 4 doses |
| 2 mg pen (yellow label) | 2 mg only | 4 doses |
NovoFine Plus 32G 4 mm needles are bundled with the pens. The pen must be primed with a "flow check" the first time it is used, but not on subsequent doses. [13] The Ozempic label allows the dosing day to be changed when needed: the next injection must be at least 48 hours after the previous one, after which the new weekly schedule begins. The 0.25 / 0.5 mg pen is the only Ozempic pen that delivers two different dose strengths from the same cartridge; the 1 mg and 2 mg pens are fixed-dose, so a patient stepping from 0.5 mg to 1 mg switches to a different pen rather than dialing the existing one to a higher number. [4]
The Ozempic label specifically allows providers to use the 1 mg dose for patients with type 2 diabetes and chronic kidney disease who have completed at least 4 weeks at 0.5 mg, regardless of A1C. This reflects the FLOW cardiorenal outcomes program, which showed renal benefit at the 1 mg dose. The 2 mg dose is reserved for patients who need additional A1C reduction beyond what 1 mg delivers and is not the routine first-line maintenance choice. [4]
Wegovy dosing schedule (semaglutide for weight management)
Wegovy uses the same molecule as Ozempic but escalates higher and follows a strict five-step monthly schedule. Skipping a step is not part of the label. Each strength comes in a single-dose, pre-mixed pen rather than a multi-dose pen. [6]
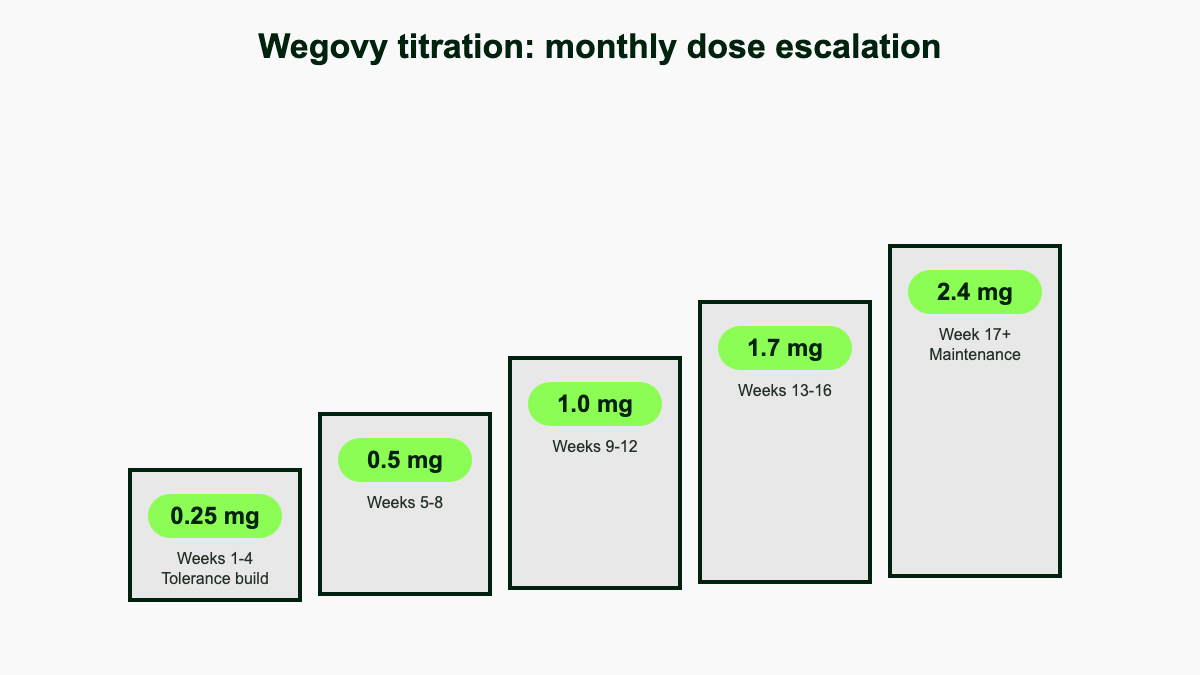
| Weeks | Weekly dose | Notes |
|---|---|---|
| 1–4 | 0.25 mg | Initiation, not effective for weight loss |
| 5–8 | 0.5 mg | Step-up |
| 9–12 | 1.0 mg | Step-up |
| 13–16 | 1.7 mg | Step-up; can serve as maintenance if 2.4 mg is not tolerated |
| 17 and ongoing | 2.4 mg | Standard maintenance |
| Optional, after ≥4 weeks at 2.4 mg | 7.2 mg (Wegovy HD) | For adults who tolerate 2.4 mg and need additional weight reduction |
The 7.2 mg pen, marketed as Wegovy HD, was approved by the FDA in March 2026 under the National Priority Voucher pilot program. It is intended only for adults who have tolerated the 2.4 mg dose for at least 4 weeks and for whom further weight reduction is clinically indicated. In the STEP UP phase 3 program, mean weight loss at 7.2 mg was 20.7 percent of body weight. [14]
Wegovy is approved for adults and for adolescents aged 12 years and older with obesity, using the same titration. If a patient cannot tolerate 2.4 mg, the label permits dropping back to 1.7 mg for one month and re-attempting. If 1.7 mg is also not tolerated, the medication should be discontinued rather than maintained at a sub-therapeutic dose; lower steps were not designed to deliver durable weight loss. [6]
Each Wegovy pen contains a single weekly dose. There is no multi-dose pen for Wegovy, so the prescription is written for a four-pack at the current step strength and a new prescription is needed for each escalation. The pen click count differs from Ozempic and is not user-adjustable: the patient simply confirms the strength on the label, primes once if it is the first use of that pen, and injects until the dose counter shows zero. Sites should be rotated and the pen should be stored refrigerated until first use, after which it can stay at room temperature for up to 28 days. [6]
Rybelsus dosing (oral semaglutide)
Rybelsus is the only oral GLP-1 currently on the market and the only one taken every day rather than weekly. Bioavailability of an oral peptide is intrinsically low (about 0.4 to 1 percent), and depends on a strict administration ritual: swallow the tablet whole with no more than 4 oz (about 120 mL) of plain water on an empty stomach immediately after waking, then wait at least 30 minutes before eating, drinking anything else, or taking other oral medications. Splitting, crushing or chewing the tablet destroys the SNAC absorption enhancer that makes oral semaglutide possible. [7][15]
| Phase | Dose | Duration |
|---|---|---|
| Initiation | 3 mg once daily | 30 days (tolerance only) |
| First therapeutic dose | 7 mg once daily | At least 30 days |
| Maximum | 14 mg once daily | Maintenance if needed |
The 3 mg starting dose is explicitly not effective for glycemic control. If a Rybelsus dose is missed, skip it and resume the next morning; do not double up and do not take two tablets to "stack" a higher dose. Two 7 mg tablets cannot be substituted for one 14 mg tablet because absorption is non-linear. [7]
Co-administered oral medications with a narrow therapeutic index (notably levothyroxine) need extra care because Rybelsus delays gastric emptying and may alter their absorption. The 30-minute fasting window applies to the levothyroxine itself: separate the two by the maximum interval the prescriber allows, and check TSH 4 to 6 weeks after starting Rybelsus or after any Rybelsus dose change. [15]
Mounjaro dose chart (tirzepatide for type 2 diabetes)
Mounjaro is dosed weekly by subcutaneous injection. Unlike semaglutide, every Mounjaro strength above the 2.5 mg starter is a potential maintenance dose, and the label permits stopping the titration at any of them once glycemic targets are met. Tirzepatide is unique among GLP-1 medicines because it is also a GIP agonist; the dual mechanism is why a 5 mg tirzepatide dose is roughly comparable to a 1 mg semaglutide dose for A1C reduction. [8]
| Phase | Dose | Minimum time before next step |
|---|---|---|
| Initiation | 2.5 mg weekly | 4 weeks |
| First therapeutic dose | 5 mg weekly | 4 weeks |
| Step up | 7.5 mg weekly | 4 weeks |
| Step up | 10 mg weekly | 4 weeks |
| Step up | 12.5 mg weekly | 4 weeks |
| Maximum | 15 mg weekly | Maintenance |
The label is explicit that doses should not be increased more often than every 4 weeks, and that 5, 10, and 15 mg are the prespecified maintenance options studied in SURPASS, with 7.5 and 12.5 mg available as intermediate steps for tolerance. [8] Mounjaro is supplied as single-dose KwikPens at all six strengths and as single-dose vials drawn up with a syringe. The vials cost less but require the patient to draw the dose accurately, which is why most diabetes prescribers default to the pen unless cost is the deciding factor. [16]
Mounjaro pens are stored refrigerated and warmed to room temperature for 30 minutes before injection to reduce the cold-injection sting that some patients report. Once removed from the refrigerator, an unused pen can stay at room temperature for up to 21 days. The pen is single-use: after one injection it is discarded in a sharps container regardless of how much liquid appears to remain in the cartridge, because the deliverable dose has already been measured. The 2.5 mg starter pack always contains 4 pens for the initiation month, while subsequent strengths are typically dispensed as a 4-week supply. [8]
Zepbound dose schedule (tirzepatide for chronic weight management)
Zepbound uses the same molecule, the same six dose strengths, and the same 4-week titration interval as Mounjaro. The difference is the indication and the labeled maintenance options: 5 mg, 10 mg, and 15 mg weekly are the recommended maintenance doses for weight management. [9]
| Weeks | Weekly dose |
|---|---|
| 1–4 | 2.5 mg (initiation) |
| 5–8 | 5 mg |
| 9–12 | 7.5 mg (if step-up needed) |
| 13–16 | 10 mg |
| 17–20 | 12.5 mg (if step-up needed) |
| 21 and ongoing | 15 mg (maximum maintenance) |
Zepbound also comes in single-dose vials at every approved strength (2.5, 5, 7.5, 10, 12.5 and 15 mg) sold through the LillyDirect Self Pay Pharmacy Solutions program. The vial pricing is tiered: 2.5 mg starts at about $299/month, 5 mg at $399/month, and 7.5 to 15 mg at $449/month when refilled within 45 days. The dosing schedule and maintenance options are identical to the autoinjector pens; the vial is simply a delivery format that lowers cash-pay cost. [9][17]
In December 2024, the FDA approved Zepbound as the first prescription medicine for moderate-to-severe obstructive sleep apnea (OSA) in adults with obesity. The titration is identical to weight management, but the labeled OSA maintenance doses are 10 mg or 15 mg once weekly: lower doses were not powered to demonstrate AHI improvement in the SURMOUNT-OSA trials, where 42 to 50 percent of treated participants achieved disease remission or mild non-symptomatic OSA at week 52. [18]
The OSA indication is additive: a patient with both obesity and moderate-to-severe OSA is on a single Zepbound prescription that addresses both conditions, not on two separate regimens. The trial design enrolled patients regardless of whether they were using continuous positive airway pressure (CPAP), and the AHI improvement was independent of CPAP use. The clinical implication is that Zepbound can be a primary OSA therapy in patients unable or unwilling to tolerate CPAP, but it does not replace a sleep study for diagnosis or follow-up monitoring. [18]
Saxenda and Victoza (liraglutide) daily titration
Liraglutide is the only daily injectable GLP-1. Both Saxenda (weight management) and Victoza (type 2 diabetes) escalate weekly rather than monthly because the half-life is roughly 13 hours rather than a week, so receptor adaptation is much faster. [10][11]
| Week | Saxenda (weight) | Victoza (T2D) |
|---|---|---|
| 1 | 0.6 mg daily | 0.6 mg daily |
| 2 | 1.2 mg daily | 1.2 mg daily (therapeutic) |
| 3 | 1.8 mg daily | Continue 1.2 mg or step to 1.8 mg |
| 4 | 2.4 mg daily | 1.8 mg daily (maximum) |
| 5+ | 3.0 mg daily (maintenance) | n/a (1.8 mg is Victoza max) |
Saxenda is approved for chronic weight management in adults and in adolescents aged 12 years and older who weigh more than 60 kg. If a patient cannot tolerate stepping up after a week, the label allows holding the current dose for an additional week before retrying. If 3.0 mg cannot be tolerated within 12 weeks of starting, Saxenda should be discontinued because lower doses have not shown sustained weight loss. [10]
Victoza is approved for adults and for pediatric patients aged 10 years and older with type 2 diabetes; the pediatric titration matches the adult schedule (0.6 mg for at least one week, then step to 1.2 mg, then 1.8 mg if needed). Victoza was discontinued by Novo Nordisk in 2024 in some markets, but the molecule and dosing remain available as authorized generic liraglutide. [11][19]
If you cannot tolerate stepping up
The single most common reason a GLP-1 fails is that the patient cannot get past a step-up because of nausea, vomiting or constipation. Every label provides for this. The default protocol, used across all seven products, is to hold the current well-tolerated dose for another full escalation interval (4 weeks for weekly injections, 1 week for liraglutide, 30 days for Rybelsus) before re-attempting the step. There is no need to drop back unless the current dose itself becomes intolerable. [3][4][6]
If the next dose was tried and produced unmanageable symptoms, dropping back one step is permitted on every label. For Wegovy, the explicit fallback is 1.7 mg if 2.4 mg cannot be tolerated. For Mounjaro and Zepbound, any of the five step doses can be held as a maintenance dose. For Saxenda, the label is the strictest: if 3.0 mg is not reached within 12 weeks of starting, discontinuation is recommended. [6][9][10]
Symptomatic management of GI side effects (small frequent low-fat meals, hydration, loperamide for diarrhea, polyethylene glycol for constipation, ondansetron for nausea on prescription) typically buys enough tolerance to complete the next step. Severe vomiting, abdominal pain radiating to the back, or any symptom suggestive of pancreatitis is a stop signal, not a tolerance issue. [3]
A useful rule of thumb is the 50-50 test: if a patient can eat at least 50 percent of a normal meal and is not vomiting more than once a day on the current dose by the end of the third week, escalation on schedule is reasonable. If either threshold is missed, holding the current dose for another four weeks gives the gut time to adapt without losing therapeutic momentum. Patients who have already lost meaningful weight on the current step often benefit more from holding than from pushing higher, because the next step adds proportionally less weight loss but a measurable additional GI burden. [3][20]
Missed-dose rules by drug
The window for catching up on a missed dose is shorter than most patients expect, and it differs by drug. The rule is set on the label, not by the prescriber. [1][2][12]

| Drug | Window to take the missed dose | If past the window |
|---|---|---|
| Ozempic | Within 5 days of the scheduled day | Skip and resume on the usual day |
| Wegovy | Next dose more than 48 hours away: take it; less than 48 hours away: skip | Skip; resume next scheduled dose |
| Mounjaro / Zepbound | Within 4 days; never inject two doses within 3 days of each other | Skip and resume regular weekly schedule |
| Rybelsus | No catch-up; missed doses are skipped | Take the next dose the following morning |
| Saxenda / Victoza | If >12 hours late, skip the dose | Resume next day; if >3 days missed, restart at 0.6 mg to re-titrate |
The Saxenda re-titration rule is the one most often missed: any gap of more than 3 days requires going back to 0.6 mg and walking up again, because GI tolerance is lost quickly with daily liraglutide. [10] For weekly semaglutide and tirzepatide, gaps of 2 or more weeks justify a call to the prescriber. The label position is to resume at the next scheduled dose, but many clinicians prefer to drop back one step after a 2-week or longer gap because GI tolerance does begin to fade. [3][20]
Switching between GLP-1 brands
There is no FDA-published equivalence table for switching between GLP-1 brands. The standard label position is to start the new drug at its labeled initiation dose rather than match the old maintenance dose, because cross-reactivity at the receptor and pharmacokinetic differences (especially when moving between weekly and daily dosing, or between semaglutide and tirzepatide) are not predictable on a milligram basis. Real-world practice is more nuanced. [3][12]
Most clinicians accept that a patient stable on a therapeutic semaglutide dose has already cleared the worst GI tolerance hurdle, so they start the new agent above the formal initiation step, watching for breakthrough nausea. The most commonly used informal mappings are below. None of these are FDA-approved; they are pragmatic starting points only. [20]
| Switching from | To | Common starting dose | Notes |
|---|---|---|---|
| Ozempic 0.5 mg weekly | Mounjaro / Zepbound | 2.5 mg weekly | Start at label initiation |
| Ozempic 1 mg weekly | Mounjaro / Zepbound | 2.5 or 5 mg weekly | Patient choice with prescriber |
| Ozempic 2 mg or Wegovy 2.4 mg | Mounjaro / Zepbound | 5 mg weekly | Skip 2.5 mg only if GI tolerance is excellent |
| Wegovy 0.25–0.5 mg | Zepbound | 2.5 mg weekly | Start at label initiation |
| Wegovy 1.0–1.7 mg | Zepbound | 5 mg weekly | Match therapeutic class |
| Wegovy 2.4 mg | Zepbound | 5 to 7.5 mg weekly | Lower if GI sensitive |
| Mounjaro / Zepbound 5 mg | Ozempic / Wegovy | 0.5 mg weekly | Start at first therapeutic step |
| Mounjaro / Zepbound 10–15 mg | Ozempic / Wegovy | 0.5 to 1 mg weekly | Re-titrate; semaglutide is mg-for-mg less potent |
| Saxenda 3 mg or Victoza 1.8 mg daily | Ozempic / Wegovy | 0.25 to 0.5 mg weekly | Daily-to-weekly transition; start lower |
| Rybelsus 14 mg daily | Ozempic | 0.5 mg weekly | Equivalent therapeutic step |
The transition timing is also informal. There is no obligatory washout: most prescribers restart on the day the next dose would have been due (one week after the last weekly injection, one day after the last Saxenda or Rybelsus dose). Waiting two weeks adds nothing, and waiting longer risks losing GI tolerance, after which the new agent has to be started at full initiation. [20]
Renal, hepatic and special-population dosing
No dose adjustment is required for any approved GLP-1 in renal impairment, including end-stage renal disease for semaglutide and tirzepatide. The label caveat is shared across all products: dehydration from vomiting or diarrhea during initiation or escalation can precipitate acute kidney injury or worsen chronic renal failure. Patients with eGFR < 60 mL/min/1.73 m² should have renal function monitored at every titration step and during any prolonged GI flare, with extra attention to volume status. [4][6][8]
Hepatic impairment also does not require a dose change for any of the seven products, but clinical experience in severe hepatic impairment is limited and the labels recommend caution. There is no GLP-1 contraindication on the basis of liver enzymes alone, but new or worsening transaminitis after starting therapy should prompt a workup. [4][8]
Pediatric dosing is currently limited to three products. Wegovy is approved for adolescents aged 12 years and older with obesity, using the same titration as adults. Saxenda is approved for adolescents aged 12 years and older who weigh more than 60 kg. Victoza is approved for pediatric type 2 diabetes from age 10 and older with the same dose schedule as adults. Ozempic, Mounjaro, Zepbound and Rybelsus remain adult-only at this time. [6][10][11]
Pregnancy and lactation are managed conservatively. Because semaglutide and tirzepatide both have very long half-lives (about 7 days for semaglutide and 5 days for tirzepatide), the labels recommend stopping the medication at least 2 months before a planned pregnancy for semaglutide and at least 4 weeks for tirzepatide. Liraglutide can be discontinued the day pregnancy is confirmed because it clears within a few days. None of the GLP-1 receptor agonists are recommended during breastfeeding because animal data show transfer into milk and human data are absent. [21]
Compounded semaglutide and tirzepatide dosing
Compounded GLP-1s prepared by 503A pharmacies follow the same general titration logic as the FDA-approved products, but the dosing is not standardized and depends entirely on the prescriber and pharmacy. Compounded semaglutide is typically supplied at concentrations of 2.5, 5 or 10 mg/mL in multi-dose vials, and the patient draws each weekly dose with an insulin syringe. Compounded tirzepatide is usually 17 or 20 mg/mL because the doses are larger by mass. [22]
Most compounded protocols mirror the FDA escalation: 0.25 mg weekly for semaglutide or 2.5 mg weekly for tirzepatide for the first 4 weeks, then a step every 4 weeks. The flexibility of vial-and-syringe dosing means many prescribers use intermediate steps not available with manufactured pens (for example 0.4 mg or 0.75 mg of semaglutide, or 3.5 or 6 mg of tirzepatide) for patients who hit a tolerance wall on the standard ramp. The trade-off is that compounded products are not FDA-approved, batch-to-batch potency depends on the pharmacy's quality controls, and FDA enforcement actions in 2024 and 2025 have narrowed the legitimate use case to clinical scenarios where the FDA-approved drug is unavailable or unsuitable for a specific patient. [22]
Compounded products labeled with additives (B12, B6, levocarnitine, glycine) do not change the GLP-1 dosing schedule, and there is no clinical evidence that the additives modify weight loss or GI tolerance. Patients moving from a compounded product to a brand-name pen typically restart at the FDA initiation dose if the compounded preparation has been off for more than 14 days, or step in at the closest manufactured strength if continuity is maintained. Document the compounded concentration and weekly dose at the time of transition so the brand-name prescriber can match it accurately. [22]
Storage and timing details that affect a single dose
Pen storage is part of dosing because temperature exposure changes the effective dose delivered. All weekly GLP-1 pens (Ozempic, Wegovy, Mounjaro, Zepbound) are refrigerated between 36 and 46 degrees F until first use. After first use or after the pen leaves the refrigerator, the labels permit storage at controlled room temperature for a defined window: 56 days for Ozempic, 28 days for Wegovy, 21 days for Mounjaro, and 21 days for Zepbound. Pens exposed to temperatures above 86 degrees F or frozen at any point should be discarded because the peptide can denature and lose potency without any visible change to the liquid. [4][6][8][9]
Daily pens (Saxenda, Victoza) follow the same refrigeration rule, with a room-temperature in-use window of 30 days. Rybelsus tablets are stored in the original blister pack at room temperature and are sensitive to humidity; the SNAC absorption enhancer degrades if the tablet is removed from the blister more than a few minutes before swallowing. [10][11][15]
Time of day for weekly injections is flexible because steady-state plasma concentration changes very little within a 24-hour window. Time of day for daily Saxenda and Victoza is also flexible, but should be consistent week-to-week so that GI symptoms peak at predictable times. Rybelsus is the exception: the morning empty-stomach window is non-negotiable because food in the stomach within 30 minutes of the tablet reduces absorption to clinically insignificant levels. [4][7][10]
How long to stay at maintenance and when to consider dose reduction
None of the GLP-1 labels define a fixed duration for maintenance therapy. Weight management products (Wegovy, Zepbound, Saxenda) are explicitly indicated for chronic use and the SELECT, SURMOUNT and SCALE Maintenance trials all show that stopping the drug leads to roughly two-thirds regain of lost weight within a year. Diabetes products (Ozempic, Mounjaro, Rybelsus, Victoza) are continued indefinitely as long as A1C targets are met and the medication is tolerated. The expected pattern is lifelong therapy with periodic dose reassessment, not a fixed treatment course followed by a taper. [3][20]
Dose reduction is appropriate in three scenarios. First, if a patient on a higher dose develops new GI intolerance after months of stability (often triggered by an unrelated illness or antibiotic course), dropping back one step for 4 to 8 weeks usually restores tolerance without losing efficacy. Second, if a patient on Zepbound or Wegovy approaches a target body weight and wants to maintain rather than lose, the lower maintenance options (5 mg Zepbound, 1.7 mg Wegovy) are reasonable provided the lower dose holds the weight loss; in practice, many patients discover they regain on the lower step and have to return to the previous strength. Third, in patients who develop sarcopenia or unexpected lean mass loss, lowering the dose and emphasizing protein intake and resistance training can reduce the appetite-suppressing effect enough to allow caloric intake to recover without losing weight benefit. [3]
Discontinuation should be planned, not abrupt. The semaglutide and tirzepatide half-lives mean that the drug remains active for 4 to 6 weeks after the last injection, so the appetite-suppressing effect tapers over that window rather than stopping the day the prescription ends. Patients planning to stop benefit from a structured plan that increases protein intake, locks in resistance training, and addresses regain proactively in the first 90 days. [20]
References
- U.S. Food and Drug Administration. Ozempic (semaglutide) injection, prescribing information. accessdata.fda.gov/drugsatfda_docs/label/2025/209637s025lbl
- U.S. Food and Drug Administration. Mounjaro (tirzepatide) injection, prescribing information. accessdata.fda.gov/drugsatfda_docs/label/2022/215866s000lbl
- MedlinePlus. Semaglutide injection. National Library of Medicine. medlineplus.gov/druginfo/meds/a618008
- Drugs.com. Semaglutide dosage: usual adult dose for diabetes type 2. drugs.com/dosage/semaglutide
- Novo Nordisk. Ozempic 2 mg approval announcement, March 2022.
- U.S. Food and Drug Administration. Wegovy (semaglutide) injection, prescribing information, including 2023 pediatric supplement. accessdata.fda.gov/drugsatfda_docs/label/2023/215256s007lbl
- Mayo Clinic. Semaglutide (oral route) description and dosing. mayoclinic.org/drugs-supplements/semaglutide-oral-route
- U.S. Food and Drug Administration. Mounjaro full prescribing information, dosage and administration section.
- U.S. Food and Drug Administration. Zepbound (tirzepatide) injection, prescribing information. pi.lilly.com/us/zepbound-uspi
- U.S. Food and Drug Administration. Saxenda (liraglutide) injection 3 mg, prescribing information. accessdata.fda.gov/drugsatfda_docs/label/2023/206321s016lbl
- U.S. Food and Drug Administration. Victoza (liraglutide) injection, prescribing information. accessdata.fda.gov/drugsatfda_docs/label/2023/022341s039lbl
- MedlinePlus. Tirzepatide injection. National Library of Medicine. medlineplus.gov/druginfo/meds/a622044
- Novo Nordisk. How to use Ozempic pen. ozempic.com/how-to-take/ozempic-dosing
- U.S. Food and Drug Administration. FDA approves higher dose semaglutide (Wegovy 7.2 mg) under National Priority Voucher Program, March 2026.
- U.S. Food and Drug Administration. Rybelsus (semaglutide) tablets, prescribing information. accessdata.fda.gov/drugsatfda_docs/label/2024/213051s018lbl
- Eli Lilly. Mounjaro KwikPen and single-dose vial product information.
- Eli Lilly. Zepbound single-dose vials available through LillyDirect Self Pay Pharmacy Solutions, investor release.
- U.S. Food and Drug Administration. FDA approves first medication for obstructive sleep apnea (Zepbound), December 2024. fda.gov/news-events/press-announcements/fda-approves-first-medication-obstructive-sleep-apnea
- DailyMed. Victoza (liraglutide injection) label. National Library of Medicine. dailymed.nlm.nih.gov
- NiceRx. Switching from semaglutide to tirzepatide dose conversion chart. nicerx.com/blog/switching-from-semaglutide-to-tirzepatide-dose-conversion-chart
- Second Nature. Wegovy and chronic kidney disease, pregnancy and lactation guidance based on FDA label. secondnature.io
- DiversifyRx. Compounded semaglutide and tirzepatide pharmacy guide for 503A practice. diversifyrx.com